(g/mo)
Your answer
0 This is a required question
Using the balanced chemical equation...

(g/mo)
Your answer
0 This is a required question
Using the balanced chemical equation below, how many moles of methane 1 point
will react to produce 3.2 moles of water? *
** Answer in numerical numbers only. Do not include units. (answer MUST include the correct
number of significant figures for *ANY* credit)
CH4 + 20, → CO2 + 2H,0
Your answer._
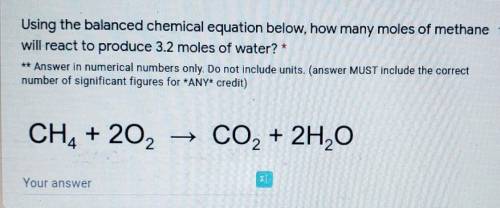

Answers: 2
Another question on Chemistry


Chemistry, 22.06.2019 12:00
What is the lowest number energy level where a d sublevel is found
Answers: 1

Chemistry, 22.06.2019 18:00
Mercury turns to vapor at 629.88 k how much heat is lost 175 g of mercury vapor at 650 current condenses to a liquid at 297 ca mercury turns to weber at 629.88 kelvin how much he is lost 175 g of mercury vapor and 650 coming condensers to liquidate 297 kevin
Answers: 2

You know the right answer?
Questions


Mathematics, 29.06.2019 02:30

Biology, 29.06.2019 02:30

English, 29.06.2019 02:30




Mathematics, 29.06.2019 02:30


Mathematics, 29.06.2019 02:30


Biology, 29.06.2019 02:30

History, 29.06.2019 02:30


English, 29.06.2019 02:30


History, 29.06.2019 02:30





