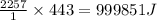Chemistry, 18.02.2020 06:27 rickyortega72701
As an athlete exercises, sweat is produced and evaporated to help maintain a proper body temperature. On average, an athlete loses approximately 443 g of sweat during an hour of exercise. How much energy is needed to evaporate the sweat that is produced? The heat of vaporization for water is 2257 J/g.

Answers: 2
Another question on Chemistry

Chemistry, 22.06.2019 07:00
Indicate whether the specified alkyl halides will form primarily substitution products, only elimination products, both substitution and elimination products, or no products when they react with sodium methoxide. 1-bromobutane 1-bromo-2-methylpropane 2-bromobutane 2-bromo-2-methylpropane
Answers: 2

Chemistry, 22.06.2019 11:00
The number to the right of an element's symbol (ex. c-12) identifies the of an isotope.
Answers: 1


Chemistry, 22.06.2019 12:10
Consider the reaction: n2(g) + o2(g) ⇄ 2no(g) kc = 0.10 at 2000oc starting with initial concentrations of 0.040 mol/l of n2 and 0.040 mol/l of o2, calculate the equilibrium concentration of no in mol/l how would this be done?
Answers: 3
You know the right answer?
As an athlete exercises, sweat is produced and evaporated to help maintain a proper body temperature...
Questions

Mathematics, 25.03.2020 22:37

Mathematics, 25.03.2020 22:37

English, 25.03.2020 22:37

Mathematics, 25.03.2020 22:37



Chemistry, 25.03.2020 22:38

Mathematics, 25.03.2020 22:38

Biology, 25.03.2020 22:38

Mathematics, 25.03.2020 22:38

Mathematics, 25.03.2020 22:38



History, 25.03.2020 22:38

Mathematics, 25.03.2020 22:38


Mathematics, 25.03.2020 22:38


Biology, 25.03.2020 22:38