
Chemistry, 17.02.2020 17:26 sedilei1515
Two equilibrium reactions of nitrogen with oxygen, with their corresponding equilibrium constants (Kc) at a certain temperature, are given below. reaction (1): N2(g) + O2(g) 2 NO(g); Kc = 1.54e-31 reaction (2): N2(g) + 1/2 O2(g) N2O(g); Kc = 2.61e-24 Using this set of data, determine the equilibrium constant for the following reaction, at the same temperature. reaction (3): N2O(g) + 1/2 O2(g) 2 NO(g)

Answers: 3
Another question on Chemistry

Chemistry, 21.06.2019 15:00
1) how many electrons are in each energy level of the following elements? a. he b. na c. na d. ne 2) how many valence electrons are percent in the following atoms? a. s b. mg c. be d. cl 3) which of the following elements are stable as atoms? a. he b. o c. cl d. ar if you are able to provide the work as to how you got the answers that would be greatly appreciated. : )
Answers: 1

Chemistry, 22.06.2019 01:00
Water is important for the of cells. a: size, shape, and temperature b: temperature, color, and odor c: color, odor, and size d: shape, temperature, and color
Answers: 2


Chemistry, 22.06.2019 14:20
Which of the following are sources of revenue for media companies? a. direct sales to producers b.advertising and subscriptions c. online purchase d. capital investments
Answers: 1
You know the right answer?
Two equilibrium reactions of nitrogen with oxygen, with their corresponding equilibrium constants (K...
Questions


Arts, 26.02.2021 18:50



Mathematics, 26.02.2021 18:50

English, 26.02.2021 18:50

Geography, 26.02.2021 18:50

History, 26.02.2021 18:50

Mathematics, 26.02.2021 18:50

Mathematics, 26.02.2021 18:50

Mathematics, 26.02.2021 18:50

Mathematics, 26.02.2021 18:50



English, 26.02.2021 18:50

Social Studies, 26.02.2021 18:50



Mathematics, 26.02.2021 18:50

Mathematics, 26.02.2021 18:50

 ; Kc = 1.54e - 31
; Kc = 1.54e - 31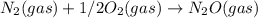 ; Kc = 2.16e - 24
; Kc = 2.16e - 24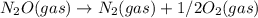 Kc = 1/2.16e - 24
Kc = 1/2.16e - 24 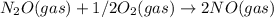 Kc = 1.54e-31 × 1/2.61e - 24
Kc = 1.54e-31 × 1/2.61e - 24

