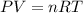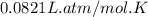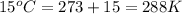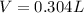Answers: 1
Another question on Chemistry

Chemistry, 21.06.2019 21:30
The reaction q+r2=r2q is found to be first order in r2 and
Answers: 1

Chemistry, 22.06.2019 05:00
Which position represents spring in the southern hemisphere? a) b) c) d)
Answers: 2

Chemistry, 22.06.2019 09:30
What are scientists who study fossils called? ( a ) astronomers. ( b ) biologists. ( c ) geologists. ( d ) paleontologists.
Answers: 2

Chemistry, 22.06.2019 11:50
Which of the following statements about hybrid orbitals is or are true? choose all that apply. choose all that apply. under sp2 hybridization, the large lobes point to the vertices of an equilateral triangle. after an atom undergoes sp hybridization there is one unhybridized p orbital on the atom. the angle between the large lobes of sp3 hybrids is 109.5∘
Answers: 2
You know the right answer?
What volume of F2 (in liters) is required to react with 1.00 g of uranium according to the equation...
Questions


History, 05.02.2020 21:46


History, 05.02.2020 21:46

Health, 05.02.2020 21:46


English, 05.02.2020 21:46

History, 05.02.2020 21:46

English, 05.02.2020 21:46



Biology, 05.02.2020 21:46





Mathematics, 05.02.2020 21:46



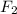 required is, 0.304 L
required is, 0.304 L
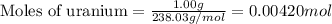
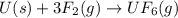
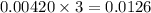 moles of
moles of