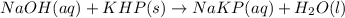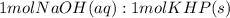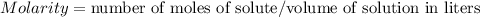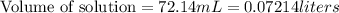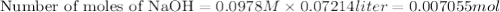Chemistry, 17.02.2020 06:06 asdf334asdf334
Answer the following questions based on the reaction below: NaOH(aq) + KHP(s) --> NaKP(aq)+H2O(I)
A 1.864 g sample of impure KHP was titrated with a 0.0978 M solution of NaOH. To completely react the KHP in the sample, 72.14 mL of base was needed. KHP (potassium hydrogen phthalate, 204.23 g/mol)
a.) How many grams of KHP were in the unknown sample?
b.) What is the percentage of KHP in the unknown sample?

Answers: 3
Another question on Chemistry

Chemistry, 22.06.2019 19:10
Astudent completes a titration by adding 12.0 milliliters of naoh(aq) of unknown concentration to 16.0 milliliters of 0.15 m hcl(aq). what is the molar concentration of the naoh(aq)? 1)5.0 m 2)0.20 m 3)0.11 m 4)1.1 m
Answers: 1

Chemistry, 23.06.2019 00:30
What would be the original temperature of a gas that has a volume of 2.0 l and a pressure of 2.0 atm and an unknown temperature that the volume increased to 3.5 l in its pressure decreased to 1.0 atm if the final temperature is measured to be 11°c
Answers: 1

Chemistry, 23.06.2019 03:30
27 drag each label to the correct location on the image. a particular exosolar system has five planets in total: a, b, c, d, and e. the table lists the orbital periods of these planets in days. planet orbital period (days) a 600 b 80 c 1,000 d 500 e 100 move each planet to its orbit in the system.
Answers: 3

You know the right answer?
Answer the following questions based on the reaction below: NaOH(aq) + KHP(s) --> NaKP(aq)+H2O(I)...
Questions



Spanish, 26.02.2021 08:30



Spanish, 26.02.2021 08:30

Mathematics, 26.02.2021 08:30


Physics, 26.02.2021 08:30



Health, 26.02.2021 08:30

Mathematics, 26.02.2021 08:30

Biology, 26.02.2021 08:30

Mathematics, 26.02.2021 08:30

Mathematics, 26.02.2021 08:30

Mathematics, 26.02.2021 08:30


Biology, 26.02.2021 08:30

Mathematics, 26.02.2021 08:30