

Answers: 2
Another question on Chemistry

Chemistry, 22.06.2019 02:50
Consider the equilibrium system: 2icl(s) ⇄ i2(s) + cl2(g) which of the following changes will increase the total amount of of cl2 that can be produced? all of the listed answers are correct decreasing the volume of the container removing the cl2 as it is formed adding more icl(s) removing some of the i2(s)
Answers: 1

Chemistry, 22.06.2019 12:00
What does a complete balanced chemical equation include? a. exothermic coefficients b. endothermic coefficients c. valence electrons d. molar coefficients
Answers: 1

Chemistry, 22.06.2019 14:30
Consider the reduction reactions and their equilibrium constants. cu+(aq)+e−↽−−⇀cu(s)pb2+(aq)+2e−↽−−⇀pb(s)fe3+(aq)+3e−↽−−⇀fe(=6.2×108=4.0×10−5=9.3×10−3 cu + ( aq ) + e − ↽ − − ⇀ cu ( s ) k =6.2× 10 8 pb 2 + ( aq ) +2 e − ↽ − − ⇀ pb ( s ) k =4.0× 10 − 5 fe 3 + ( aq ) +3 e − ↽ − − ⇀ fe ( s ) k =9.3× 10 − 3 arrange these ions from strongest to weakest oxidizing agent.
Answers: 3

Chemistry, 22.06.2019 14:30
In water, a strong acid will break down into its component parts. a. completely b. partly c. never in water, a weak base will break down into its component parts. a. completely b. partly c. never
Answers: 2
You know the right answer?
How do you find the limiting reactant if 2.50 mol copper and 5.50 mol silver nitrate react by single...
Questions


Mathematics, 04.03.2021 18:50


History, 04.03.2021 18:50



Mathematics, 04.03.2021 18:50

Mathematics, 04.03.2021 18:50

Biology, 04.03.2021 18:50

Social Studies, 04.03.2021 18:50


Mathematics, 04.03.2021 18:50

Mathematics, 04.03.2021 18:50


Chemistry, 04.03.2021 18:50

Chemistry, 04.03.2021 18:50

History, 04.03.2021 18:50

Mathematics, 04.03.2021 18:50


Mathematics, 04.03.2021 18:50

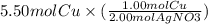 = 2.75 mol Cu.
= 2.75 mol Cu.

