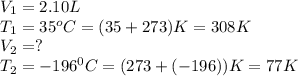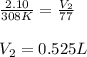Chemistry, 15.02.2020 04:44 youngbeauty17
A balloon is filled with He gas to a volume of 2.10 L at 35 °C. The balloon is placed in liquid nitrogen until its temperature reaches –196 °C. Assuming the pressure remains constant, what is the volume of the cooled balloon?

Answers: 2
Another question on Chemistry

Chemistry, 22.06.2019 06:00
Calculate - analysis of compound composed of iron and oxygen yields 174.86 of fe and 75.14g of o. what is the empirical formula for this compound?
Answers: 3

Chemistry, 22.06.2019 07:40
The formation of a solid, also known as a is an indication of a chemical change. precipitate particulate particle powder
Answers: 3

Chemistry, 22.06.2019 15:00
Which substance is a steroid? cholesterol fatty acid monosaccharide trans fat
Answers: 1

Chemistry, 22.06.2019 18:00
To apply in a gold the individual gold atoms are united to each other by means of a metallic bond. how would you use the gold block to determine the atomic radius of a gold atom?
Answers: 3
You know the right answer?
A balloon is filled with He gas to a volume of 2.10 L at 35 °C. The balloon is placed in liquid nitr...
Questions


Medicine, 09.02.2021 03:00


Mathematics, 09.02.2021 03:00


Mathematics, 09.02.2021 03:00

English, 09.02.2021 03:00

Mathematics, 09.02.2021 03:00

English, 09.02.2021 03:00

Mathematics, 09.02.2021 03:00


Mathematics, 09.02.2021 03:00

Law, 09.02.2021 03:00

Mathematics, 09.02.2021 03:00

English, 09.02.2021 03:00

Mathematics, 09.02.2021 03:00



Chemistry, 09.02.2021 03:00

History, 09.02.2021 03:00

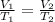
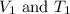 are the initial volume and temperature of the gas.
are the initial volume and temperature of the gas.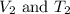 are the final volume and temperature of the gas.
are the final volume and temperature of the gas.