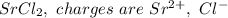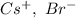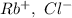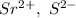Chemistry, 14.02.2020 19:38 alicemareus
The charges and sizes of the ions in an ionic compound affect the strength of the electrostatic attraction holding that compound together. Based on ion charges and relative ion sizes, rank these ionic compounds by their expected melting points from highest to lowest
a. SrCl2 ,b. CsBr ,c. RbCl ,d. SrS

Answers: 2
Another question on Chemistry

Chemistry, 22.06.2019 07:00
How far is the region from the equator or control climate
Answers: 1

Chemistry, 22.06.2019 10:00
Ahydrogen atom has 1 electron. how many bonds can hydrogen form? a) 1 b) 2 c) 3 d) 4 e) 5
Answers: 3

Chemistry, 22.06.2019 12:00
the mississippians were considered to be horticulturalists, which means they were
Answers: 1

Chemistry, 22.06.2019 20:40
Select the correct value for the indicated bond angle in each of the compounds. o−o−oo−o−o angle of o3 90° 109.5° < 109.5° 120° < 120° 180° f−b−ff−b−f angle of bf3 180° < 109.5° < 120° 120° 109.5° 90° f−o−ff−o−f angle of of2 < 120° 120° 90° 109.5° 180° < 109.5° cl−be−clcl−be−cl angle of becl2 90° 109.5° 180° 120° < 109.5° < 120° f−p−ff−p−f angle of pf3 90° 109.5° < 109.5° 180° 120° < 120° h−c−hh−c−h angle of ch4 90° < 109.5° 180° 120° < 120° 109.5°
Answers: 1
You know the right answer?
The charges and sizes of the ions in an ionic compound affect the strength of the electrostatic attr...
Questions


Health, 28.02.2020 20:51


Mathematics, 28.02.2020 20:51


Biology, 28.02.2020 20:51




Chemistry, 28.02.2020 20:51


Social Studies, 28.02.2020 20:51

Biology, 28.02.2020 20:51


Mathematics, 28.02.2020 20:51


History, 28.02.2020 20:51


Mathematics, 28.02.2020 20:51

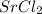 > RbCl > CsBr
> RbCl > CsBr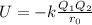
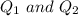 are charges and
are charges and 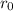 is the size.
is the size.