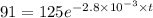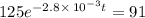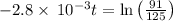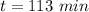The decomposition of dinitrogen pentoxide, N2O5, to NO2 and O2 is a first-order reaction. At 60°C, the rate constant is 2.8 × 10-3min-1. If a rigid vessel initially contains only N2O5 at a pressure of 125 kPa, how long will it take for the total pressure to reach 176 kPa?
a. 113 min
b. 129 min
c. 42 min
d. 182 min
e. 62 min
f. 83 min

Answers: 3
Another question on Chemistry

Chemistry, 21.06.2019 20:40
If equal masses of the listed metals were collected , which would have a greatest volume ? a. aluminum 2.70,b.zinc7.14,c.copper 8.92,d.lead 11.34
Answers: 2


Chemistry, 22.06.2019 09:00
Which explanation is true about what happens to a ray of light when it strikes a mirror? a. a light ray is transmitted toward a mirror at a certain angle. the light ray is then reflected by the mirror at an equal angle but in the opposite direction of the transmitted ray. b. an incident ray strikes a mirror at an angle with a line perpendicular to the mirror. the light ray is then reflected at an angle equal to the angle of incidence but on the opposite side of the perpendicular line. c. a reflecting ray strikes a mirror at an angle with a line perpendicular to the mirror. the light ray is then refracted at an angle equal to the angle of the reflecting ray and on the same side of the perpendicular line. d. an incident ray strikes a mirror at an angle with a line parallel to the mirror. the light ray is then transmitted at an angle equal to the angle of incidence but on the opposite side of the parallel line. you so much! : -d take the time to try and answer correctly.
Answers: 3

Chemistry, 22.06.2019 12:30
The bond energy for the van der waals bond between two helium atoms is 7.9×10−4ev. assuming that the average kinetic energy of a helium atom is (3/2)kbt, at what temperature is the average kinetic energy equal to the bond energy between two helium atoms
Answers: 1
You know the right answer?
The decomposition of dinitrogen pentoxide, N2O5, to NO2 and O2 is a first-order reaction. At 60°C, t...
Questions

Mathematics, 21.01.2020 07:31

History, 21.01.2020 07:31


Mathematics, 21.01.2020 07:31

Mathematics, 21.01.2020 07:31


Social Studies, 21.01.2020 07:31

Computers and Technology, 21.01.2020 07:31


Chemistry, 21.01.2020 07:31



Health, 21.01.2020 07:31

Mathematics, 21.01.2020 07:31


English, 21.01.2020 07:31




![[A_t]=[A_0]e^{-kt}](/tpl/images/0510/6499/1ef89.png)
![[A_t]](/tpl/images/0510/6499/5262c.png) is the concentration at time t
is the concentration at time t
![[A_0]](/tpl/images/0510/6499/9a686.png) is the initial concentration
is the initial concentration
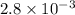 min⁻¹
min⁻¹