
Chemistry, 13.02.2020 22:21 viktoria1198zz
Assume that the reaction for the formation of gaseous hydrogen fluoride from hydrogen and fluorine has an equilibrium constant of 1.15 X 102 at a certain temperature. In a particular experiment, 3.00 mole of each component was added to a 1.50 L flask. Calculate the equilibrium concentrations of all species.

Answers: 1
Another question on Chemistry

Chemistry, 22.06.2019 09:30
Apump contains 0.5 l of air at 203 kpa.you draw back on the piston of the pump, expanding the volume until the pressure reads 50.8 kpa. what is the new volume of the air pump
Answers: 2

Chemistry, 22.06.2019 13:00
6. using 3 – 4 sentences explain (in your own words) why water expands when it freezes? 7. using your knowledge of colligative properties explain whether sodium chloride or calcium chloride would be a more effective substance to melt the ice on a slick sidewalk. use 3 – 4 sentences in your explanation.
Answers: 1

Chemistry, 23.06.2019 01:30
Polar bears give birth and hunt on sea ice. which of the following would polar bears survive during the melting of arctic ice? growing another layer of fur during summer migrate inland to search for different food sources staying put until the ice refreezes sticking to the usual diet of seals
Answers: 1

Chemistry, 23.06.2019 03:00
What happens in the particles of a gas when the gas is compressed
Answers: 1
You know the right answer?
Assume that the reaction for the formation of gaseous hydrogen fluoride from hydrogen and fluorine h...
Questions


World Languages, 23.08.2019 14:30

Mathematics, 23.08.2019 14:30



Biology, 23.08.2019 14:30

History, 23.08.2019 14:30



Social Studies, 23.08.2019 14:30

Mathematics, 23.08.2019 14:30

Computers and Technology, 23.08.2019 14:30

Mathematics, 23.08.2019 14:30


History, 23.08.2019 14:30

Computers and Technology, 23.08.2019 14:30

Biology, 23.08.2019 14:30



Mathematics, 23.08.2019 14:30

![[H_2]= 0.314 M](/tpl/images/0510/5228/1fffd.png)
![[F_2]=0.314 M](/tpl/images/0510/5228/5de3f.png)
![[HF]=3.372 M](/tpl/images/0510/5228/8e291.png)
![[H_2]=\frac{3.00 mol}{1.5 L}=2.0 M](/tpl/images/0510/5228/f9117.png)
![[F_2]=\frac{3.00 mol}{1.5 L}=2.0 M](/tpl/images/0510/5228/b656e.png)
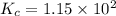
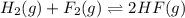
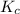 is given by :
is given by :![K_c=\frac{[HF]^2}{[H_2][F_2]}](/tpl/images/0510/5228/a2854.png)
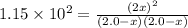
![[H_2]=(2.0 -1.686)M = 0.314 M](/tpl/images/0510/5228/64b44.png)
![[F_2]=(2.0 -1.686)M = 0.314 M](/tpl/images/0510/5228/f4a49.png)
![[HF]=(2\times 1.686)M = 3.372 M](/tpl/images/0510/5228/dc9b5.png)


