Chemistry, 13.02.2020 18:40 llamasking
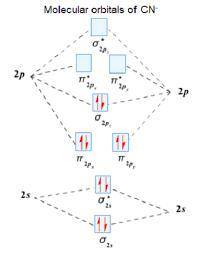
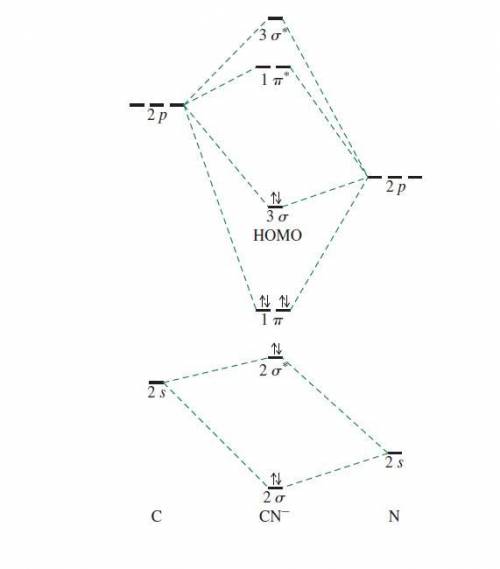
Using the molecular orbital model, write electron configurations for the following diatomic species and calculate the bond orders. Which ones are paramagnetic? Place the species in order of increasing bond length and bond energy.
a. CN +
b. CN
c. CN -

Answers: 3
Another question on Chemistry

Chemistry, 21.06.2019 13:00
What is the h+ concentration for an aqueous solution with poh = 4.01 at 25 ∘c? express your answer to two significant figures and include the appropriate units.
Answers: 1

Chemistry, 21.06.2019 20:30
9. write the chemical equation for the following word equations. include symbols for physical states in the equation. a. solid zinc sulfide + oxygen gas -> solid zinc oxide + sulfur dioxide gas b. aqueous hydrochloric acid + aqueous barium hydroxide -> aqueous barium chloride + water
Answers: 1

Chemistry, 21.06.2019 23:30
Calculate the expected ph values of the buffer systems from the experiments (a,b,c,d), using the henderson- hasselbalch equation, ph-pka+log[a-]/[ha]. use for pka values carbonic acid= 6.37, and acetic acid= 4.75.
Answers: 2

Chemistry, 22.06.2019 14:30
According to le chatelier’s principle, a system in chemical equilibrium responds to stress by shifting the equilibrium in a direction that reduces the stress. normalizes the stress. increases the stress. changes the stress.
Answers: 1
You know the right answer?
Using the molecular orbital model, write electron configurations for the following diatomic species...
Questions


Biology, 28.01.2020 03:31




Chemistry, 28.01.2020 03:31

Social Studies, 28.01.2020 03:31

Mathematics, 28.01.2020 03:31

Social Studies, 28.01.2020 03:31



Mathematics, 28.01.2020 03:31

Chemistry, 28.01.2020 03:31


Mathematics, 28.01.2020 03:31

History, 28.01.2020 03:31


Chemistry, 28.01.2020 03:31


Health, 28.01.2020 03:31