
Chemistry, 13.02.2020 05:55 jasonoliva13
Which of the following sets are isoelectronic (i. e., have the same number of electrons)? i. Br-, Kr, Sr2+ ii. C, N-, O2- iii. Mg2+, Ca2+, Sr2+ iv. O2-, O, O2+ v. Ag+, Cd2+, Pd

Answers: 3
Another question on Chemistry

Chemistry, 22.06.2019 00:40
During which time interval does the object travel approximately 10 meters
Answers: 3

Chemistry, 22.06.2019 14:00
The two naturally occurring isotopes of chlorine are 35cl (34.969 amu, 75.77%) and 37cl (36.966 amu, 24.23%). the two naturally occurring isotopes of bromine are 79br (78.918 rm amu, 50.69%) and 81br (80.916 amu, 49.31%). chlorine and bromine combine to form bromine monochloride, brcl. 1. how many peaks will be present in a mass spectrum for brcl? the four combinations of molecule possible given these four isotopes are: 81br37cl, 81br35cl, 79br37cl, and 79br35cl. 2. what are the masses of the four different brcl molecules? express the masses using six significant figures, in decreasing numeric order (highest to lowest), separated by commas.
Answers: 3

Chemistry, 22.06.2019 18:00
How many moles of oxygen gas are produced from the decomposition of six moles of potassium
Answers: 1

Chemistry, 22.06.2019 22:00
All of the following are homogeneous mixtures except a) sugar dissolved in water. b) orange juice. c) coffee with cream. d) household vinegar. e) apple juice
Answers: 1
You know the right answer?
Which of the following sets are isoelectronic (i. e., have the same number of electrons)? i. Br-, Kr...
Questions

Mathematics, 04.09.2020 22:01


Social Studies, 04.09.2020 22:01

English, 04.09.2020 22:01


Mathematics, 04.09.2020 22:01

English, 04.09.2020 22:01




Business, 04.09.2020 22:01

Mathematics, 04.09.2020 22:01



Mathematics, 04.09.2020 22:01

Mathematics, 04.09.2020 22:01


Mathematics, 04.09.2020 22:01

History, 04.09.2020 22:01

Mathematics, 04.09.2020 22:01

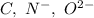
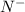 has 6 electrons.
has 6 electrons.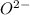 has 6 electrons.
has 6 electrons.

