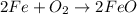A student reacts steel wool with oxygen by touching the fibers with a 9 volt battery. They begin
with 7.93 g Fe and measure the mass of the final product at 9.50 g.
The balanced equation for the reaction is:
2 Fe + 02 --> 2 FeO
A) The student claims that the percent yield of the reaction was 93.1 %. Support or reject their
claim including a calculation of percent yield as part of your evidence.

Answers: 1
Another question on Chemistry

Chemistry, 22.06.2019 06:30
(1.6 × 10-19)(5.0 × 106) = c × 10d identify the missing numbers below to show the result of multiplying the numbers.
Answers: 1


Chemistry, 22.06.2019 20:00
Acm ruler with main graduations from 1 to 10 from left to right there are 10 secondary graduations between each of the main graduations there is a line that begins. at the left end of the ruler 10 secondary graduations to the left of the “1 main graduation the right end of the line ends on the eighth secondary graduation to the right of 3 how long is the line
Answers: 1

Chemistry, 22.06.2019 21:00
What is the chemical formula for the compound formed between sodium and flour one
Answers: 1
You know the right answer?
A student reacts steel wool with oxygen by touching the fibers with a 9 volt battery. They begin
Questions


Mathematics, 02.01.2020 10:31

Chemistry, 02.01.2020 10:31




Biology, 02.01.2020 10:31

Business, 02.01.2020 10:31

Chemistry, 02.01.2020 10:31

Mathematics, 02.01.2020 10:31

Mathematics, 02.01.2020 10:31

Mathematics, 02.01.2020 10:31


History, 02.01.2020 10:31

Biology, 02.01.2020 10:31



Mathematics, 02.01.2020 10:31

History, 02.01.2020 10:31