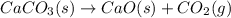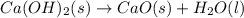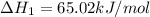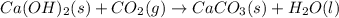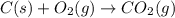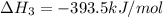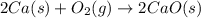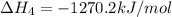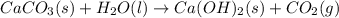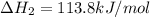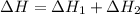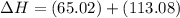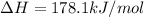Chemistry, 12.02.2020 23:10 makayladurham19
Determine the enthalpy change for the decomposition of calcium carbonate. CaCO3 (s) --> CaO (s) + CO2 (g) given the thermochemical equations below:
Ca(OH)2 (s) --> CaO (s) + H2O (l) enthalpy reaction = 65.2 kJ/mol
Ca(OH)2 (s) + CO2(g) --> CaCO3 (s) + H2O (l) enthalpy reaction = -113.8 kJ/mol
C(s) + O2 (g) --> CO2 (g) enthalpy of reation = -393.5 kJ/mol
2Ca(s) + O2(g) --> 2 CaO (s) enthalpy of reaction = -1270.2 kJ/mol
a. 1711.7 kJ/mol rxn
b. 441 kJ/mol rxn
c. 179 kJ/mol rxn
d. 48 kJ/mol rxn
e. 345.5 kJ. mol rxn

Answers: 3
Another question on Chemistry

Chemistry, 21.06.2019 22:30
Which statement best describes the oxidation numbers of the atoms found in magnesium chloride? a. magnesium has a 2- oxidation number and chlorine has a 1+ oxidation number. b. magnesium has a 2- oxidation number and chlorine has a 2+ oxidation number. c. magnesium has a 2+ oxidation number and chlorine has a 1- oxidation number. d. magnesium has a 1+ oxidation number and chlorine has a 1- oxidation number.
Answers: 2

Chemistry, 22.06.2019 04:30
What are the primary responsibilities of a chemical engineer involved in "r& d"? develop large scale manufacturing operations discover new products and processes training of new chemists determine products needed by consumers
Answers: 2

Chemistry, 22.06.2019 09:00
If you chip a tooth, most likely you will go to the dentist to have the missing material filled in. currently the material used to fill in teeth is a polymer that is flexible when put in, yet is hardened to the strength of a tooth after irradiation with blue light at a wavelength of 461 nm. what is the energy in joules for a photon of light at this wavelength?
Answers: 1

Chemistry, 22.06.2019 12:00
An atom's configuration based on its number of electrons ends at 3p4. another atom has seven more electrons. starting at 3p, what is the remaining configuration? 3p63d34s2 3p43d54s2 3p64s23d3 3p44s23d
Answers: 3
You know the right answer?
Determine the enthalpy change for the decomposition of calcium carbonate. CaCO3 (s) --> CaO (s) +...
Questions

Social Studies, 18.08.2019 17:30

Physics, 18.08.2019 17:30

Mathematics, 18.08.2019 17:30

Social Studies, 18.08.2019 17:30

Social Studies, 18.08.2019 17:30


Physics, 18.08.2019 17:30

Chemistry, 18.08.2019 17:30


Mathematics, 18.08.2019 17:30






English, 18.08.2019 17:30


Mathematics, 18.08.2019 17:30


Mathematics, 18.08.2019 17:30