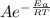Chemistry, 12.02.2020 05:30 dededese2403
Which of the following statements is TRUE?a. The rate constant does not depend on the activation energy for a reaction where the products are lower in energy than the reactants. b. The addition of a homogeneous catalyst does not change the activation energy of a given reaction. c. Rate constants are temperature dependent. d. None of the above is true. e. A catalyst raises the activation energy of a reaction.

Answers: 2
Another question on Chemistry


Chemistry, 22.06.2019 08:30
The mass of a neutron is equal to the mass of a proton plus the mass of an electron. true or false false true
Answers: 1


You know the right answer?
Which of the following statements is TRUE?a. The rate constant does not depend on the activation ene...
Questions

Mathematics, 17.09.2020 14:01

Mathematics, 17.09.2020 14:01

Spanish, 17.09.2020 14:01

English, 17.09.2020 14:01

Mathematics, 17.09.2020 14:01

Mathematics, 17.09.2020 14:01

Biology, 17.09.2020 14:01

Mathematics, 17.09.2020 14:01

Mathematics, 17.09.2020 14:01

Mathematics, 17.09.2020 14:01

Mathematics, 17.09.2020 14:01

Mathematics, 17.09.2020 14:01

Mathematics, 17.09.2020 14:01

Mathematics, 17.09.2020 14:01

Mathematics, 17.09.2020 14:01

French, 17.09.2020 14:01

Physics, 17.09.2020 14:01

Health, 17.09.2020 14:01

Mathematics, 17.09.2020 14:01

History, 17.09.2020 14:01