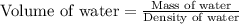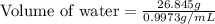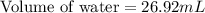Chemistry, 12.02.2020 04:41 carlosgc19
A student obtained a clean, dry, glass-stoppered flask. He weighed the flask stopper and found the total mass to be 32.634g. He then filled the flask with water, weighed again, and obtained a mass of 59.479g. At the temperature of the water, he found that its density was 0.9973 g/mL. a.) What was the mass of the water? (show work)b.) What was the volume of the water? (Show work)c.) What was the volume of the flask? (show work)

Answers: 1
Another question on Chemistry

Chemistry, 22.06.2019 12:40
Consider the directing effects of the substituents on salicylamide and predict the possible structures of the iodination products. which do you think will be the major product?
Answers: 1

Chemistry, 22.06.2019 22:00
Choose all the answers that apply. fluorine (f) has an atomic number of 9 and an atomic weight of 18.99. fluorine has a. 9 protons b. 10 neutrons c. 18 electrons d. an atomic mass of 19 e. at least one isotope
Answers: 1


Chemistry, 23.06.2019 02:30
Apound is approximately 0.45 kilogram. a persons weighs 87 kilograms. what is the persons’s weight, in pounds, when expressed to the correct number of significant figures
Answers: 1
You know the right answer?
A student obtained a clean, dry, glass-stoppered flask. He weighed the flask stopper and found the t...
Questions

Chemistry, 14.05.2021 03:40



Chemistry, 14.05.2021 03:40

Mathematics, 14.05.2021 03:40


Mathematics, 14.05.2021 03:40




Mathematics, 14.05.2021 03:40


Social Studies, 14.05.2021 03:40


Biology, 14.05.2021 03:40

Mathematics, 14.05.2021 03:40


Mathematics, 14.05.2021 03:40

English, 14.05.2021 03:40

Mathematics, 14.05.2021 03:40