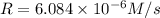The initial rate data at 25 oC are listed for the reaction
NH4+ (aq) + NO2-1 -> N2(g) + H2O(l)
Experiment Initial [NH4+1] Initial [NO2-1] Initial rate (M/s)
1 0.24 0.1 7.2 X10^-6
2 0.12 0.1 3.6 X 10^-6
3 0.12 0.15 5.4 X 10^-6
a. Determine the rate law
b. Determine the value of the rate constant.
c. What is the reaction rate when the concentrations are [NH4+1] = 0.39 M and [NO2-1] = 0.052 M.

Answers: 3
Another question on Chemistry

Chemistry, 22.06.2019 08:30
For each of the compounds below, show that the charges on the ions add up to zero. a. kbr b. cao c. li(2)o d. cacl(2) e. alcl(3)
Answers: 2

Chemistry, 23.06.2019 05:30
How many moles are in 1.26*10^24 particles in significant figures
Answers: 2

Chemistry, 23.06.2019 11:30
The density of e85 fuel is 0.801 g/ml. what is the mass of 1.00 gallon of the fuel? (1 gal. = 3.785 l)
Answers: 3

Chemistry, 23.06.2019 14:30
Which statement best identifies the process shown? the process must be fusion because energy is released. a.the process must be fusion because a heavier nucleus forms from smaller nuclei. b.the process must be fission because a large nucleus breaks into smaller nuclei. c.the process must be fission because neutrons are formed.
Answers: 1
You know the right answer?
The initial rate data at 25 oC are listed for the reaction
NH4+ (aq) + NO2-1 -> N2(g)...
NH4+ (aq) + NO2-1 -> N2(g)...
Questions

Mathematics, 18.09.2020 21:01

Mathematics, 18.09.2020 21:01

Physics, 18.09.2020 21:01

Mathematics, 18.09.2020 21:01

Social Studies, 18.09.2020 21:01

Mathematics, 18.09.2020 21:01

Mathematics, 18.09.2020 21:01

Mathematics, 18.09.2020 21:01

Mathematics, 18.09.2020 21:01

History, 18.09.2020 21:01

English, 18.09.2020 21:01

Mathematics, 18.09.2020 21:01

Mathematics, 18.09.2020 21:01

Mathematics, 18.09.2020 21:01

Mathematics, 18.09.2020 21:01

English, 18.09.2020 21:01

Mathematics, 18.09.2020 21:01

Mathematics, 18.09.2020 21:01

Mathematics, 18.09.2020 21:01

Mathematics, 18.09.2020 21:01

![=k[NH_4^{+}]^1[NO_2^{-}]^1](/tpl/images/0507/8247/fe0cf.png)
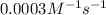
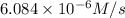 is the reaction rate when the concentrations are
is the reaction rate when the concentrations are ![[NH_4^{+}]](/tpl/images/0507/8247/7aa39.png) = 0.39 M and
= 0.39 M and ![[NO_2^{-}]](/tpl/images/0507/8247/f8e12.png) = 0.052 M.
= 0.052 M.![=k[NH_4^{+}]^x[NO_2^{-}]^y](/tpl/images/0507/8247/8a532.png)
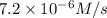
![R=k[0.24 M]^x[0.1 M]^y](/tpl/images/0507/8247/5fc35.png) ...[1]
...[1]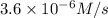
![R'=k[0.12 M]^x[0.1 M]^y](/tpl/images/0507/8247/447c7.png) ...[2]
...[2]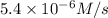
![R''=k[0.12 M]^x[0.15 M]^y](/tpl/images/0507/8247/eedcf.png) ...[3]
...[3]![\frac{R}{R'}=\frac{k[0.24 M]^x[0.1]^y}{k[0.12 M]^x[0.1]^y}](/tpl/images/0507/8247/5b2fa.png)
![\frac{7.2\times 10^{-6} M/s}{3.6\times 10^{-6} M/s}=\frac{k[0.24 M]^x[0.1 M]^y}{k[0.12 M]^x[0.1 M]^y}](/tpl/images/0507/8247/5d06d.png)
![\frac{R'}{R''}=\frac{k[0.12 M]^x[0.1 M]^y}{k[0.12 M]^x[0.15]^y}](/tpl/images/0507/8247/52502.png)
![\frac{3.6\times 10^{-6} M/s}{5.4\times 10^{-6} M/s}=\frac{k[0.12 M]^x[0.1 M]^y}{k[0.12 M]^x[0.15 M]^y}](/tpl/images/0507/8247/db163.png)
![7.2\times 10^{-6} M/s=k[0.24 M]^1[0.1 M]^1](/tpl/images/0507/8247/29f30.png) ...[1]
...[1]![k=\frac{7.2\times 10^{-6} M/s}{[0.24 M]^1[0.1 M]^1}=0.0003 M^{-1} s^{-1}](/tpl/images/0507/8247/38560.png)
![R=0.0003 M^{-1} s^{-1}\times [0.39 M][0.052 M]](/tpl/images/0507/8247/2f5df.png)