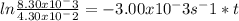The conversion of methyl isonitrile to acetonitrile in the gas phase at 250 °C CH3NC(g)CH3CN(g) is first order in CH3NC with a rate constant of 3.00×10-3 s-1. If the initial concentration of CH3NC is 4.30×10-2 M, the concentration of CH3NC will be 8.30×10-3 M after s have passed.

Answers: 1
Another question on Chemistry

Chemistry, 22.06.2019 05:00
Given sno2 + 2h2 - sn + 2h20 tin oxide reacts with hydrogen to produce tin and water. how many moles of sno2 are needed to produce 500.0 grams of sn?
Answers: 3

Chemistry, 22.06.2019 08:30
Analyze how limestone is weathered and identify the features that are formed as a result of this dissolution
Answers: 1


Chemistry, 22.06.2019 17:30
Consider the story you just read. all but one of the choices below indicate that something is living.
Answers: 1
You know the right answer?
The conversion of methyl isonitrile to acetonitrile in the gas phase at 250 °C CH3NC(g)CH3CN(g) is f...
Questions




Mathematics, 11.02.2020 05:02




Social Studies, 11.02.2020 05:02

Computers and Technology, 11.02.2020 05:02




Social Studies, 11.02.2020 05:03


Computers and Technology, 11.02.2020 05:03


English, 11.02.2020 05:03



Mathematics, 11.02.2020 05:03

![ln \frac{[A]_t}{[A]_0} = -kt](/tpl/images/0507/3637/bde68.png)
![[A]_t](/tpl/images/0507/3637/82372.png) is the concentration of the reactant in the time t,
is the concentration of the reactant in the time t, ![[A]_0](/tpl/images/0507/3637/7075c.png) is the initial concentration of the reactant, k is rate constant and t is time. Replacing with values of the problem:
is the initial concentration of the reactant, k is rate constant and t is time. Replacing with values of the problem: