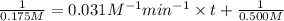Chemistry, 11.02.2020 04:29 2018jecr46871
Based on the data given and a rate constant of 0.031 M?1?min?1, calculate the time at which the concentration of reactant A will be 0.175M .
t (min) [A]t(M)
0.00 0.500
20.0 0.382
40.0 0.310
60.0 0.260
80.0 0.2

Answers: 2
Another question on Chemistry

Chemistry, 21.06.2019 22:30
1. combine iron and copper (ii) sulfate solution. (hint: iron will form the iron (iii) ion) fe + cuso4 → 2. combine lead (ii) nitrate and potassium iodide solutions. pb(no3)2+ kl → 3. combine magnesium metal and hydrochloric acid solution. mg + hcl → 4. electrolysis (splitting) of water. h2o → 5. burning magnesium. mg + o2 →
Answers: 3

Chemistry, 22.06.2019 02:20
Brainliesttt me asap! the below reaction can be categorized as more than one type of reaction. which reactions are these, and what are the types of reactions?
Answers: 1

Chemistry, 22.06.2019 04:00
The rules of engagement (roe) working group is often used to (select all that apply.)
Answers: 2

Chemistry, 22.06.2019 05:30
What type of reaction is shown below? check all that apply. 2h2o2 → 2h2o + o2 synthesis decomposition combustion
Answers: 1
You know the right answer?
Based on the data given and a rate constant of 0.031 M?1?min?1, calculate the time at which the conc...
Questions

Social Studies, 03.10.2021 19:10


English, 03.10.2021 19:10

Mathematics, 03.10.2021 19:10

English, 03.10.2021 19:10

English, 03.10.2021 19:10




Mathematics, 03.10.2021 19:10

Advanced Placement (AP), 03.10.2021 19:10


Mathematics, 03.10.2021 19:10


Mathematics, 03.10.2021 19:10


Chemistry, 03.10.2021 19:10

English, 03.10.2021 19:10

Mathematics, 03.10.2021 19:10

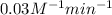
![[A_o]=0.50M](/tpl/images/0506/1572/e4719.png)
![[A]=0.175 M](/tpl/images/0506/1572/c5f24.png)
![\frac{1}{[A]}=kt+\frac{1}{[A_0]}](/tpl/images/0506/1572/4158d.png)