
Chemistry, 11.02.2020 04:25 Andychulo7809
Calculate the amount of heat (in kJ) required to convert 62.6 g of water to steam at 100°C. (The molar heat of vaporization of water is 40.79 kJ/mol.)

Answers: 1
Another question on Chemistry


Chemistry, 22.06.2019 10:10
When water dissociates, each water molecule splits into a hydroxide ion and a) h 3 o + b) a hydrogen atom c) a hydrogen ion d) h 2 o e) oh —
Answers: 2

Chemistry, 22.06.2019 10:40
Ammonia and oxygen react to form nitrogen monoxide and water, like this: 4nh3 (g) + 5o2 (g) → 4no (g) + 6h2o (g) also, a chemist finds that at a certain temperature the equilibrium mixture of ammonia, oxygen, nitrogen monoxide, and water has the following composition: compound pressure at equilibrium nh3 65.1atm o2 31.3atm no 62.7atm h2o 65.8atm compound pressure at equilibrium nh3 65.3 atm o2 7.79 atm no 12.1 atm h2o 65.8 atm calculate the value of the equilibrium constant kp for this reaction. round your answer to 2 significant
Answers: 2

Chemistry, 22.06.2019 15:00
Phosphorous acid, h3po3(aq) , is a diprotic oxyacid that is an important compound in industry and agriculture. p k a1 p k a2 1.30 6.70 calculate the ph for each of the points in the titration of 50.0 ml of 1.5 m h3po3(aq) 1.5 m h 3 po 3 ( aq ) with 1.5 m koh(aq). 1.5 m koh ( aq ) .
Answers: 1
You know the right answer?
Calculate the amount of heat (in kJ) required to convert 62.6 g of water to steam at 100°C. (The mol...
Questions




Mathematics, 25.04.2020 12:45


History, 25.04.2020 12:46

Biology, 25.04.2020 12:48



History, 25.04.2020 12:49


Mathematics, 25.04.2020 12:50

Mathematics, 25.04.2020 12:51

History, 25.04.2020 12:52

History, 25.04.2020 12:52

Biology, 25.04.2020 12:54

Mathematics, 25.04.2020 12:54


Mathematics, 25.04.2020 12:56

Biology, 25.04.2020 12:57

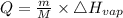
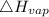 = 40.79 kJ/mol
= 40.79 kJ/mol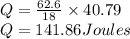
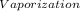 ·m where Q = required heat energy,
·m where Q = required heat energy, 


