
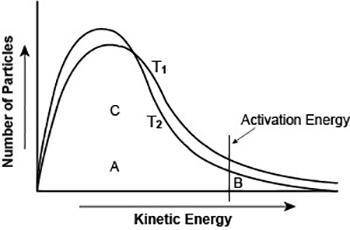
The graph shows the distribution of energy in the particles of two gas samples at different temperatures, T1 and T2. A, B, and C represent individual particles.
Based on the graph, which of the following statements is likely to be true? (3 points)
Particle A is more likely to participate in the reaction than particle B.
Particle C is more likely to participate in the reaction than particle B.
The number of particles able to undergo a chemical reaction is less than the number that is not able to.
More gas particles participate in the reaction at T2 than at T1.

Answers: 3
Another question on Chemistry

Chemistry, 22.06.2019 08:00
Why is the bond angle in a water molecule less than the bond angle of methane? a. the central oxygen atom in water has two lone pairs of electrons, whereas the central carbon atom in methane has no lone pairs. b. the central hydrogen atom in water has one lone pair of electrons, whereas the central carbon atom in methane has two lone pairs. c. the central oxygen atom in water has four lone pairs of electrons, whereas the central carbon atom in methane has only one lone pair. d. the central oxygen atom exerts more repulsive force on surrounding atoms than the central carbon atom in methane does. reset next
Answers: 2

Chemistry, 22.06.2019 09:30
Based on its chemical properties, identify the position of each chemical family on the periodic table.
Answers: 3

Chemistry, 22.06.2019 14:00
Will mark brainliest how many electrons can be held in the energy level n = 4?
Answers: 1

Chemistry, 22.06.2019 16:00
Sulfuric acid is a polyprotic acid. write balanced chemical equations for the sequence of reactions that sulfuric acid can undergo when it's dissolved in water.
Answers: 2
You know the right answer?
The graph shows the distribution of energy in the particles of two gas samples at different temperat...
Questions

History, 25.08.2019 08:30



History, 25.08.2019 08:30

Social Studies, 25.08.2019 08:30

History, 25.08.2019 08:30

Geography, 25.08.2019 08:30

Mathematics, 25.08.2019 08:30

Mathematics, 25.08.2019 08:30

English, 25.08.2019 08:30

Chemistry, 25.08.2019 08:30

Mathematics, 25.08.2019 08:30


English, 25.08.2019 08:30

English, 25.08.2019 08:30


History, 25.08.2019 08:30

Mathematics, 25.08.2019 08:30

History, 25.08.2019 08:30




