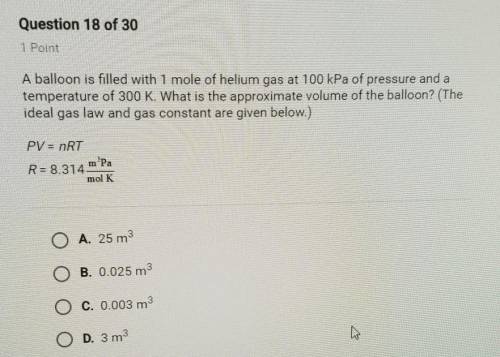
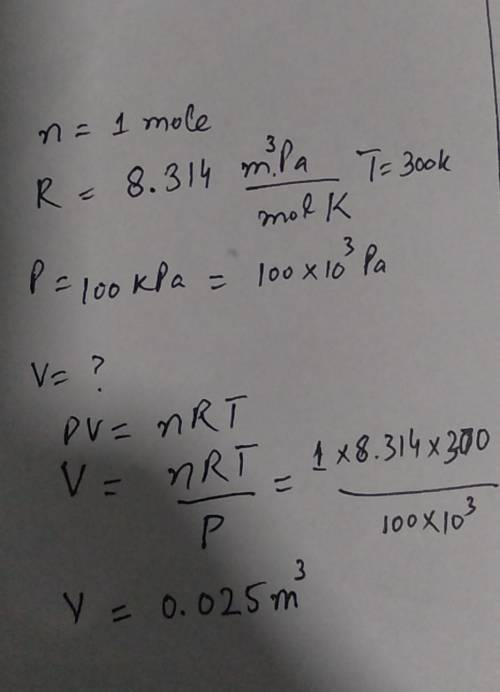A balloon is filled with 1 mole of helium gas at 100 kPa of pressure and a
temperature of 300 K...

Chemistry, 10.02.2020 21:07 cordobamariana07
A balloon is filled with 1 mole of helium gas at 100 kPa of pressure and a
temperature of 300 K. What is the approximate volume of the balloon? (The
ideal gas law and gas constant are given below.)
PV = nRT
R=8.314 m. Pa/
mol K

Answers: 1
Another question on Chemistry

Chemistry, 21.06.2019 22:00
To save time, you can approximate the initial mass of the solid to the nearest ±1 g. for example, if you are asked to add 14.3 g of copper, add between 13 g and 15 g. which of the following sets include two samples with an equal density? which all that apply below 15.4 g gold and 18.7 g silver 15.2 g copper and 50.0 g copper 20.2 g silver and 20.2 g copper 11.2 g gold and 14.9 g gold
Answers: 1

Chemistry, 22.06.2019 04:30
How much energy is made when a pice of wood burns. how do you know
Answers: 2

Chemistry, 22.06.2019 22:00
Plz ill give u brainliest which of the following steps is not likely to take place during cellular respiration? a.oxygen combines with carbon of simple sugar. b. energy molecule transfers energy to cells. d. energy is used up.
Answers: 3

You know the right answer?
Questions


English, 03.02.2020 02:53

Biology, 03.02.2020 02:53

Mathematics, 03.02.2020 02:53

English, 03.02.2020 02:53



English, 03.02.2020 02:53


Biology, 03.02.2020 02:53


Social Studies, 03.02.2020 02:53

Mathematics, 03.02.2020 02:53


Mathematics, 03.02.2020 02:53



Biology, 03.02.2020 02:53

Social Studies, 03.02.2020 02:53