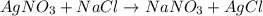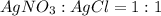The reaction below shows how silver chloride can be synthesized.
agno3 + nacl mc017-1.jpg nano...

Chemistry, 18.09.2019 23:50 cfnewton09
The reaction below shows how silver chloride can be synthesized.
agno3 + nacl mc017-1.jpg nano3 + agcl
how many moles of silver chloride are produced from 15.0 mol of silver nitrate?
1) 1.0 mol
2) 15.0 mol
3) 30.0 mol
4) 45.0 mol

Answers: 2
Another question on Chemistry

Chemistry, 21.06.2019 17:30
Write a paragraph at least 10 sentences explaining how rocks are used today in society
Answers: 1

Chemistry, 22.06.2019 01:00
Water is important for the of cells. a: size, shape, and temperature b: temperature, color, and odor c: color, odor, and size d: shape, temperature, and color
Answers: 2

Chemistry, 22.06.2019 04:40
Silver tarnishes as silver metal reacts with hydrogen sulfide, h2s, in the air. in this reaction, dark silver sulfide, au2s, covers the surface of silver. when silver is polished, this coating of silver sulfide can be removed from the surface. this makes the silver shiny again. enter the coefficients that balance the tarnishing reaction equation. (type 1 for no coefficient.)
Answers: 2

You know the right answer?
Questions

English, 09.01.2020 19:31


Chemistry, 09.01.2020 19:31


Mathematics, 09.01.2020 19:31

Mathematics, 09.01.2020 19:31

Mathematics, 09.01.2020 19:31






History, 09.01.2020 19:31


History, 09.01.2020 19:31





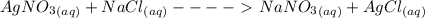
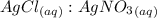 = 1 : 1
= 1 : 1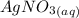 = 15.0 mol
= 15.0 mol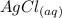 = 15.0 mol
= 15.0 mol