Chemistry, 04.02.2020 05:44 laxraAragon
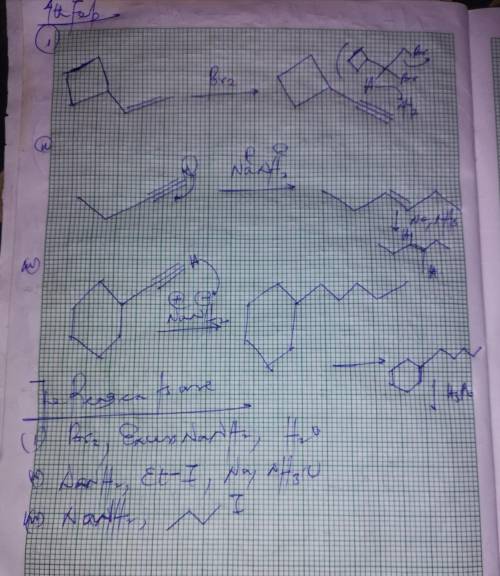
Ab c d 1) nanh2 2) mei 3) 9-bbn 4) h2o2, naoh 1) br2 2) excess nanh2 3) h2o 1) br2 2) excess nanh2 3) h2o 4) h2so4, h2o, hgso4 1) nanh2 2) eti 3) na, nh3 (l) e f g h 1) 9-bbn 2) h2o2, naoh 1) excess nanh2 2) h2o 3) br2 (1 eq), ccl4 1) excess nanh2 2) h2o 3) nanh2 4) mei 5) na, nh3 (l) 1) nanh2 2) 3) h2, pt

Answers: 3
Another question on Chemistry



Chemistry, 22.06.2019 12:00
Consider the following reaction at equilibrium. 2co2 (g) 2co (g) + o2 (g) h° = -514 kj le châtelier's principle predicts that the equilibrium partial pressure of co (g) can be maximized by carrying out the reaction a. at high temperature and high pressure b. at high temperature and low pressure c. at low temperature and low pressure d. at low temperature and high pressure e. in the presence of solid carbon
Answers: 2

Chemistry, 22.06.2019 13:00
Lab reagent, hypothesis test.a reference solution used as a lab reagent is purported to have a concentration of 5 mg/dl. six samples are taken from this solution and the following concentrations are recorded: (5.32, 4.88, 5.10, 4.73, 5.15, 4.75) mg/dl.these six measurements are assumed to be an srs of all possible measurements from solution.they are also assumed to have a standard deviation of 0.2, a normal distributin, and a mean concentration equal to the true concentration of the solution.carry out a significance test to determine whether these six measurements provide reliable evidence that the true concentration of the solution is actually not 5 mg/dl.
Answers: 1
You know the right answer?
Ab c d 1) nanh2 2) mei 3) 9-bbn 4) h2o2, naoh 1) br2 2) excess nanh2 3) h2o 1) br2 2) excess nanh2 3...
Questions


History, 25.11.2019 07:31


Geography, 25.11.2019 07:31



Social Studies, 25.11.2019 08:31

Social Studies, 25.11.2019 08:31

Biology, 25.11.2019 08:31


Business, 25.11.2019 08:31


Advanced Placement (AP), 25.11.2019 08:31

Business, 25.11.2019 08:31

Chemistry, 25.11.2019 08:31


Mathematics, 25.11.2019 08:31


Mathematics, 25.11.2019 08:31