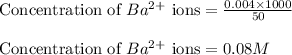Chemistry, 29.01.2020 00:48 brooke3493
A20.0 ml sample of 0.200 m k₂co₃ solution is added to 30.0 ml of 0.400 m ba(no₃)₂ solution. barium carbonate precipitates.
the concentration of barium ion, ba²⁺, in solution after the reaction is

Answers: 3
Another question on Chemistry

Chemistry, 21.06.2019 22:30
1. combine iron and copper (ii) sulfate solution. (hint: iron will form the iron (iii) ion) fe + cuso4 → 2. combine lead (ii) nitrate and potassium iodide solutions. pb(no3)2+ kl → 3. combine magnesium metal and hydrochloric acid solution. mg + hcl → 4. electrolysis (splitting) of water. h2o → 5. burning magnesium. mg + o2 →
Answers: 3


Chemistry, 22.06.2019 13:50
What happens when an atom of sulfur combines with two atoms of chlorine to produce sci2? a. each chlorine atom shares a pair of electrons with the sulfur atom. b. an electron is transferred from each chlorine atom to the sulfur atom. c. an electron is transferred from the sulfur atom to each chlorine atom. d. each chlorine atom shares all its valence electrons with the sulfur atom.
Answers: 2

Chemistry, 22.06.2019 17:30
In a heat of an engine, if 700 j enters the system, and the piston does 400 j of work what is the final internal (thermal) energy of the system if the initial energy is 1,500 j
Answers: 2
You know the right answer?
A20.0 ml sample of 0.200 m k₂co₃ solution is added to 30.0 ml of 0.400 m ba(no₃)₂ solution. barium c...
Questions

Mathematics, 07.10.2020 06:01

Business, 07.10.2020 06:01

Mathematics, 07.10.2020 06:01

Mathematics, 07.10.2020 06:01

Mathematics, 07.10.2020 06:01

Computers and Technology, 07.10.2020 06:01



English, 07.10.2020 06:01

Mathematics, 07.10.2020 06:01


Mathematics, 07.10.2020 06:01





Physics, 07.10.2020 06:01

English, 07.10.2020 06:01


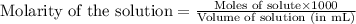 .....(1)
.....(1)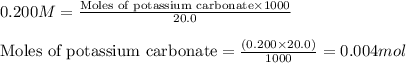
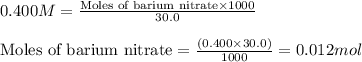
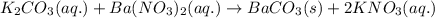
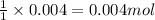 of barium nitrate
of barium nitrate