
Chemistry, 28.01.2020 06:31 yarielperez5788
Which of the following aqueous solutions contains the greatest number of ions?
a. 400. ml of 0.10 m nacl;
b. 300. ml of 0.10 m cacl2;
c. 200. ml of 0.10 m fecl3

Answers: 2
Another question on Chemistry


Chemistry, 22.06.2019 11:50
Calculate the molarity of each of the following solutions. part a) 0.12 mol of lino3 in 5.5 l of solution part b) 60.7 g c2h6o in 2.48 l of solution part c) 14.2 mg ki in 100 ml of solution
Answers: 2

Chemistry, 22.06.2019 13:00
16. why must the number of electrons lost equal the number of electrons gained in every redox reaction? use 3 – 4 sentences in your own words to address this question. 18. what type of radiation is emitted when chromium-51 decays into manganese-51? show the nuclear equation that leads you to this answer. 19. a radioactive nucleus alpha decays to yield a sodium-24 nucleus in 14.8 hours. what was the identity of the original nucleus? show the nuclear equation that leads you to this answer.
Answers: 2

Chemistry, 22.06.2019 20:00
Iam hoping to create 5.72 grams of glucose. the plant was given 4.75 liters of co2 and 2.81 g of h20. which reactant was the limiting reagent? how much excess mass did we have of the other reactant?
Answers: 1
You know the right answer?
Which of the following aqueous solutions contains the greatest number of ions?
a. 400....
a. 400....
Questions



Mathematics, 02.09.2020 04:01

Mathematics, 02.09.2020 04:01

Mathematics, 02.09.2020 04:01


Computers and Technology, 02.09.2020 04:01


Biology, 02.09.2020 04:01







Mathematics, 02.09.2020 04:01

Chemistry, 02.09.2020 04:01



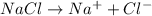
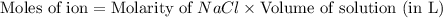
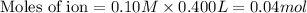
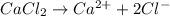
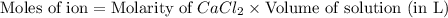
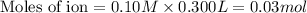
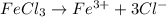
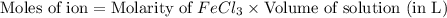
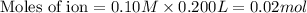
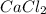 aqueous solutions contains the greatest number of ions.
aqueous solutions contains the greatest number of ions.

