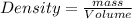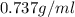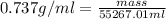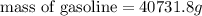Density is an intensive physical property that relates the mass of an object to its volume. density, which is simply the mass of an object divided by its volume, is expressed in the si derived unit g/mlg/ml for a liquid or g/cm3g/cm3 for a solid. most substances expand or contract when heated or cooled, so the density values for a substance are temperature dependent. a particular brand of gasoline has a density of 0.737 g/ml at 25 ? c. how many grams of this gasoline would fill a 14.6gal tank?

Answers: 3
Another question on Chemistry

Chemistry, 22.06.2019 06:10
Explain the relationship between forward and backward reactions in equilibrium, and predict how changing the amount of a reactant (creating a tension) will affect that relationship.
Answers: 1


Chemistry, 22.06.2019 19:20
For a research project, a student decided to test the effect of the lead(ii) ion (pb2+) on the ability of salmon eggs to hatch. this ion was obtainable from the water‐soluble salt, lead(ii) nitrate, which the student decided to make by the following reaction. pbo(s) + 2 hno3(aq) → pb(no3)2(aq) + h2o losses of product for various reasons were expected, and a yield of 86.0% was expected. in order to have 5.00 g of product at this yield, how many grams of pbo should be reacted? (assume that sufficient nitric acid, hno3, would be used.)
Answers: 1

Chemistry, 22.06.2019 21:30
The solid xy decomposes into gaseous x and y: xy(s) m x(g) + y(g) kp = 4.1 (at 0 °c) if the reaction is carried out in a 22.4 l container, which initial amounts of x and y will result in the formation of solid xy?
Answers: 1
You know the right answer?
Density is an intensive physical property that relates the mass of an object to its volume. density,...
Questions

History, 07.10.2020 08:01




Mathematics, 07.10.2020 08:01






Mathematics, 07.10.2020 08:01


Mathematics, 07.10.2020 08:01



Chemistry, 07.10.2020 08:01