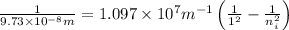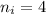Chemistry, 27.01.2020 23:31 xxtonixwilsonxx
Ahydrogen atom in an excited state emits a photon of frequency ν = 3.084 x 1015 s-1. if the electron returns to the ground state, in which level was it before the photon was emitted?

Answers: 1
Another question on Chemistry

Chemistry, 22.06.2019 02:30
At 40 âc the solution has at 40 â c the solution has blank g of k n o 3 per 100 g of water and it can contain up to blank g of k n o 3 per 100 g of water. at 0 â c the solubility is ~ blank g k n o 3 per 100 g of water, so ~ blank g k n o 3 per 100 g of water will precipitate out of solution.g of kno3 per 100 g of water and it can contain up to at 40 â c the solution has blank g of k n o 3 per 100 g of water and it can contain up to blank g of k n o 3 per 100 g of water. at 0 â c the solubility is ~ blank g k n o 3 per 100 g of water, so ~ blank g k n o 3 per 100 g of water will precipitate out of solution.g of kno3 per 100 g of water. at 0 âc the solubility is ~ at 40 â c the solution has blank g of k n o 3 per 100 g of water and it can contain up to blank g of k n o 3 per 100 g of water. at 0 â c the solubility is ~ blank g k n o 3 per 100 g of water, so ~ blank g k n o 3 per 100 g of water will precipitate out of solution.kno3 per 100 g of water, so ~ at 40 â c the solution has blank g of k n o 3 per 100 g of water and it can contain up to blank g of k n o 3 per 100 g of water. at 0 â c the solubility is ~ blank g k n o 3 per 100 g of water, so ~ blank g k n o 3 per 100 g of water will precipitate out of solution.gkno3 per 100 g of water will precipitate out of solution. a kno3 solution containing 55 g of kno3 per 100.0 g of water is cooled from 40 ∘c to 0 ∘c. what will happen during cooling?
Answers: 2


Chemistry, 22.06.2019 17:10
Acalorimeter is to be calibrated: 51.203 g of water at 55.2 degree c is added to a calorimeter containing 49.783 g of water at 23.5c. after stirring and waiting for the system to equilibrate, the final temperature reached is 37.6 degree c. specific heat capacity of water (s = 4.18 j/g∙degree c). calculate the calorimeter constant. (smδt)warm water = -[(smδt)cold water + (calorimeterδtcold water)]
Answers: 2

Chemistry, 23.06.2019 01:30
At a certain temperature the rate of this reaction is first order in hi with a rate constant of : 0.0632s2hig=h2g+i2g suppose a vessel contains hi at a concentration of 1.28m . calculate how long it takes for the concentration of hi to decrease to 17.0% of its initial value. you may assume no other reaction is important. round your answer to 2 significant digits.
Answers: 1
You know the right answer?
Ahydrogen atom in an excited state emits a photon of frequency ν = 3.084 x 1015 s-1. if the electron...
Questions


Spanish, 27.06.2019 22:40

Mathematics, 27.06.2019 22:40

Mathematics, 27.06.2019 22:40


Health, 27.06.2019 22:40

Mathematics, 27.06.2019 22:40



Mathematics, 27.06.2019 22:40


Biology, 27.06.2019 22:40




Computers and Technology, 27.06.2019 22:40



Computers and Technology, 27.06.2019 22:40

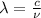
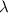 = wavelength of the light
= wavelength of the light 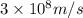
 = frequency of light =
= frequency of light = 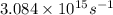
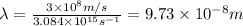
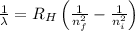
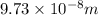
 = Rydberg's Constant =
= Rydberg's Constant = 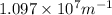
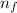 = Final energy level = 1
= Final energy level = 1 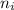 = Initial energy level = ?
= Initial energy level = ?