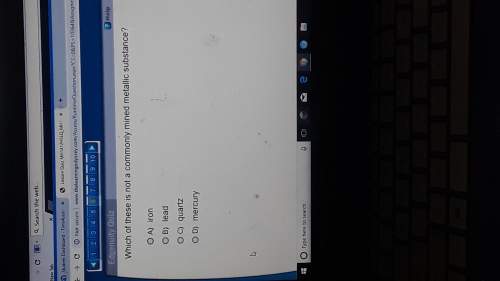
The chemical equation below shows the decomposition of ammonium nitrate (nh4no3).
nh4no3 → n2o + 2h20
a chemist who is performing this reaction starts with 160.1 g of nh4no3. the molar mass of nh4no3 is 80.03 g/mol; the molar
mass of water (h2o) is 18.01 g/mol. what mass, in grams, of h2o is produced?
9.01
18.01
36.03
7206

Answers: 2
Another question on Chemistry


Chemistry, 22.06.2019 12:00
Why are people not able to skip a dive to the deepest part of the ocean
Answers: 1

Chemistry, 22.06.2019 16:30
An atom with 7 protons, 6 neutrons, and 7 electrons has an atomic mass of amu. (enter a whole number.) numerical answers expected! answer for blank 1:
Answers: 3

Chemistry, 22.06.2019 22:30
The vapor pressure of ethanol is 1.00 × 102 mmhg at 34.90°c. what is its vapor pressure at 61.61°c? (δhvap for ethanol is 39.3 kj/mol.)
Answers: 2
You know the right answer?
The chemical equation below shows the decomposition of ammonium nitrate (nh4no3).
nh4no3 → n2o...
nh4no3 → n2o...
Questions




Health, 01.12.2020 14:10




Social Studies, 01.12.2020 14:10

Chemistry, 01.12.2020 14:10

Biology, 01.12.2020 14:10




English, 01.12.2020 14:10

Social Studies, 01.12.2020 14:10


Health, 01.12.2020 14:10



English, 01.12.2020 14:10