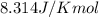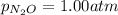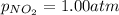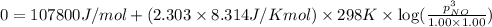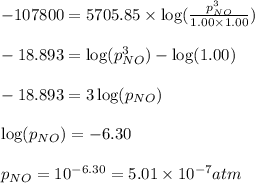Chemistry, 25.01.2020 03:31 6710000831
Consider this reaction occurring at 298 k:
n2o(g) + no2(g) ⇆ 3 no(g)
if a reaction mixture contains only n2o and no2 at partial pressures of 1.0 atm each, the reaction will be spontaneous until some no forms in the mixture.
what maximum partial pressure of no builds up before the reaction ceases to be spontaneous?

Answers: 3
Another question on Chemistry

Chemistry, 21.06.2019 19:30
The ph of carrots are 5.0 how it is classified a.acidic b.basic c.indicator d.neutral
Answers: 2

Chemistry, 21.06.2019 22:20
One or more substances changing into one or more substances is an example of a
Answers: 1

Chemistry, 22.06.2019 13:50
Read the chemical equation. 2c2h2 + 5o2 → 4co2 + 2h2o which of the following statements would be correct if one mole of c2h2 was used in this reaction? one mole of oxygen was used in this reaction. five moles of oxygen were used in this reaction. four moles of carbon dioxide were produced from this reaction. two moles of carbon dioxide were produced from this reaction.
Answers: 3

Chemistry, 22.06.2019 21:30
What is happening when the water inside a kettle heats up and begins to boil
Answers: 1
You know the right answer?
Consider this reaction occurring at 298 k:
n2o(g) + no2(g) ⇆ 3 no(g)
if a...
n2o(g) + no2(g) ⇆ 3 no(g)
if a...
Questions

Mathematics, 08.12.2020 21:10


Mathematics, 08.12.2020 21:10


Chemistry, 08.12.2020 21:10



Mathematics, 08.12.2020 21:10

Mathematics, 08.12.2020 21:10



Biology, 08.12.2020 21:10



World Languages, 08.12.2020 21:10


Medicine, 08.12.2020 21:10


Mathematics, 08.12.2020 21:10

Biology, 08.12.2020 21:10

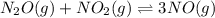
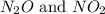 at partial pressures of 1.0 atm each, the reaction will be spontaneous until some NO forms in the mixture.
at partial pressures of 1.0 atm each, the reaction will be spontaneous until some NO forms in the mixture.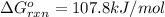
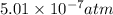
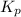 for above equation follows:
for above equation follows: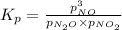
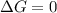 (at equilibrium)
(at equilibrium)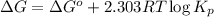
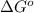 = Standard Gibbs free energy = 107.8 kJ/mol = 107800 J/mol (Conversion factor: 1 kJ = 1000 J )
= Standard Gibbs free energy = 107.8 kJ/mol = 107800 J/mol (Conversion factor: 1 kJ = 1000 J )