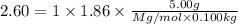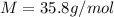Chemistry, 24.01.2020 22:31 freshysans4
The freezing point of an aqueous solution containing an unknown solute is -2.60 degc. the solution was prepared by dissolving 5.00 g of a nonelectrolytic solute in 100. ml of water. what is the molar mass of the unknown solute?

Answers: 1
Another question on Chemistry

Chemistry, 22.06.2019 02:00
If you add 10ml of hot water to 10ml of cold water and the change in tempature 8°c calculate how much energy is gained by the cold water
Answers: 1

Chemistry, 22.06.2019 08:00
Asap! will give brainiest when a heat wave strikes a region causing more people to run air-conditioning units, electrical demand increases. what needs to be done to meet this increased demand? raising the control rodslowering the control rodsremoving the control rods
Answers: 1


Chemistry, 22.06.2019 14:30
For the reaction shown, find the limiting reactant for each of the following initial amounts of reactants. 4al(s)+3o2(g)→2al2o3(s) a) 1 molal, 1 mol o2 b) 4 molal, 2.6 mol o2 c) 16 molal, 13 mol o2 d) 7.4 molal, 6.5 mol o2
Answers: 3
You know the right answer?
The freezing point of an aqueous solution containing an unknown solute is -2.60 degc. the solution w...
Questions


English, 28.09.2019 04:30

Biology, 28.09.2019 04:30

Computers and Technology, 28.09.2019 04:30

Chemistry, 28.09.2019 04:30

Social Studies, 28.09.2019 04:30


Health, 28.09.2019 04:30

Mathematics, 28.09.2019 04:30








Mathematics, 28.09.2019 04:30



Health, 28.09.2019 04:30

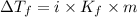
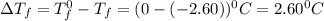 = Depression in freezing point
= Depression in freezing point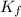 = freezing point constant =
= freezing point constant = 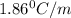
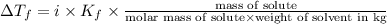
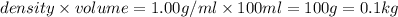 (1kg=1000g)
(1kg=1000g)