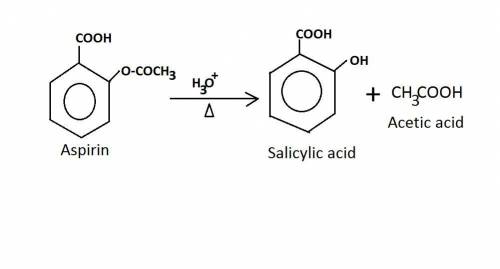
Old aspirin exposed to moisture often smells like acetic acid (vinegar). when aspirin is heated in boiling water, it decomposes and gives off a vinegar smell. the resulting solution gives a positive fecl3 test. why is this test positive? ( explain in detail! )
write the chemical equation for the reaction of aspirin and water at high temperature.

Answers: 1
Another question on Chemistry

Chemistry, 22.06.2019 02:50
What is the overall order of reaction for rate = k[no2]2 ? second order 3/2 order third order zero order none of the listed answers are correct
Answers: 3



Chemistry, 22.06.2019 19:30
Draw the lewis structure for the trisulfur s3 molecule. be sure to include all resonance structures that satisfy the octet rule.
Answers: 3
You know the right answer?
Old aspirin exposed to moisture often smells like acetic acid (vinegar). when aspirin is heated in b...
Questions

History, 09.10.2019 04:30

Spanish, 09.10.2019 04:30

Mathematics, 09.10.2019 04:30




Social Studies, 09.10.2019 04:30

Chemistry, 09.10.2019 04:30


Mathematics, 09.10.2019 04:30


Social Studies, 09.10.2019 04:30



History, 09.10.2019 04:30

Mathematics, 09.10.2019 04:30

Social Studies, 09.10.2019 04:30

Biology, 09.10.2019 04:30


Spanish, 09.10.2019 04:30

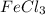 .
.