
Chemistry, 24.01.2020 21:31 vistagallosky
6. barium sulfate and and sodium sulfate react in a double displacement reaction. if the
reaction starts with 10.25 grams of barium sulfate what are the products and how many moles
of each product is produced?
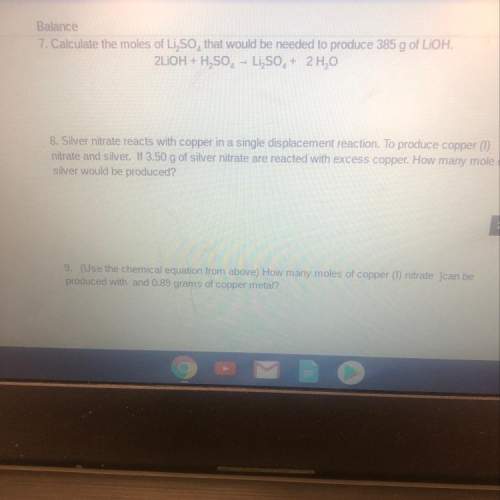
7. calculate the moles of li2so4that would be needed to produce 385 g of lioh.
8. silver nitrate reacts with copper in a single displacement reaction. to produce copper (1)
nitrate and silver. if 3.50 g of silver nitrate are reacted with excess copper. how many mole of
silver would be produced?
9. (use the chemical equation from above) how many moles of copper (1) nitrate ]can be
produced with and 0.89 grams of copper metal?

Answers: 2
Another question on Chemistry

Chemistry, 22.06.2019 03:00
Which best describes how johannes kepler developed his laws of planetary motion
Answers: 3

Chemistry, 22.06.2019 18:00
What amount of heat is exchanged when 106.2 grams of substance y goes from a liquid at 35 degrees celsius to a solid at the same temperature? melting point of substance y = 35 degrees c; δhvaporization = 3.67 j/mol; δhfusion = 3.30 j/mol. mwsubstance y = 28.22 g/mol. −12.4 j −3.51 x 102 j 1.24 x 101 j 351 j
Answers: 1

Chemistry, 22.06.2019 19:30
What is the mass of oxygen gas is consumed in a reaction that produces 4.60mol so2
Answers: 3

You know the right answer?
6. barium sulfate and and sodium sulfate react in a double displacement reaction. if the
react...
react...
Questions

Mathematics, 04.12.2020 20:50



Mathematics, 04.12.2020 20:50


History, 04.12.2020 20:50


History, 04.12.2020 20:50

Mathematics, 04.12.2020 20:50

Mathematics, 04.12.2020 20:50


Mathematics, 04.12.2020 20:50

Mathematics, 04.12.2020 20:50

Mathematics, 04.12.2020 20:50

Social Studies, 04.12.2020 20:50

Mathematics, 04.12.2020 20:50



Biology, 04.12.2020 20:50



