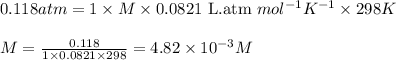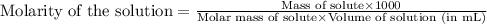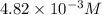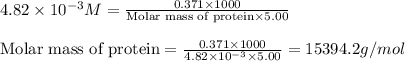Chemistry, 24.01.2020 01:31 niyyyareligion
371. mg of an unknown protein are dissolved in enough solvent to make 5.00 ml of solution. the osmotic pressure of this solution is measured to be at 0.118 atm at 25 c
calculate the molar mass of the protein.

Answers: 1
Another question on Chemistry

Chemistry, 22.06.2019 01:30
If 34.2 grams of lithium react with excess water, how many liters of hydrogen gas can be produced at 299 kelvin and 1.21 atmospheres? 2 li (s) + 2 h2o (l) yields 2 lioh (aq) + h2 (g)
Answers: 3

Chemistry, 22.06.2019 05:00
Use the table to identify the phase and phase changes of the elements under the given conditions. write the name of the substance, phase, or phase change
Answers: 3

Chemistry, 22.06.2019 08:30
The mass of a neutron is equal to the mass of a proton plus the mass of an electron. true or false false true
Answers: 1

Chemistry, 22.06.2019 14:00
The content of manganese (mn) in steel was determined spectrophotometrically and with the use of the standard addition method. an unknown sample of mn from a digested steel sample gave an absorbance of 0.185 when analyzed spectrophotometrically. when 5.00 ml of solution containing 95.5 ppm mn was added to 50.0 ml of the unknown steel solution (digested sample), the absorbance was 0.248. calculate the concentration, in parts-per-million (ppm), of mn in the digested steel sample solution.
Answers: 3
You know the right answer?
371. mg of an unknown protein are dissolved in enough solvent to make 5.00 ml of solution. the osmot...
Questions

SAT, 02.10.2019 10:30

History, 02.10.2019 10:30


History, 02.10.2019 10:30

Business, 02.10.2019 10:30





History, 02.10.2019 10:30

Biology, 02.10.2019 10:30

Biology, 02.10.2019 10:30



Social Studies, 02.10.2019 10:30

Mathematics, 02.10.2019 10:30




Mathematics, 02.10.2019 10:30

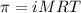
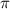 = osmotic pressure of the solution = 0.118 atm
= osmotic pressure of the solution = 0.118 atm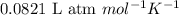
![25^oC=[273+25]=298K](/tpl/images/0468/1226/6a9f9.png)