Chemistry, 23.01.2020 05:31 lraesingleton
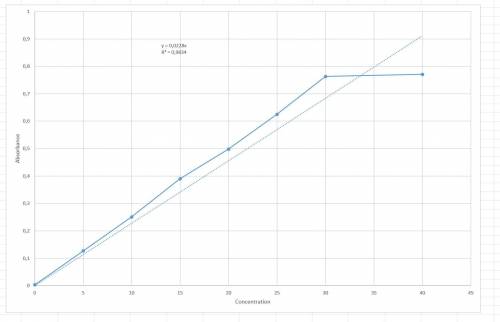
The following results were obtained when each of a series of standard silver solutions was analyzed by flame-atomic absorption spectrometry: concentration on 0 5 10 15 20 25 30 40 (ng. ml1) absorbance(r. u.) 0.003 0.127 0.251 0.390 0.498 0.625 0.763 0.771 determine the slope and intercept of the calibration plot, along with their confidence limits (95%). using the data from exercise 1 above, estimate the confidence limits for the silver concentration in: a) a sample giving an absorbance of 0.456 in a single determination. b) a sample giving absorbance values of 0.308, 0.317, 0.347, and 0.412 in four separate determinations. graph: a straight line excel plot is shown in the question

Answers: 3
Another question on Chemistry

Chemistry, 21.06.2019 18:00
Balance this equation: n2 + h2 > nh3, write the following molar ratios: n2 / n2 / nh3 h2 /
Answers: 1

Chemistry, 23.06.2019 00:00
Total the mass on the syringe. record it in the correct row of the data table. kg done click and drag weights to change the pressure. click the syringe to zoom in and see the volume. intro
Answers: 3

Chemistry, 23.06.2019 01:00
Which of the following is the molecular formula for a simple sugar? a. cooh b. h2o c. oh d. c6h12o6
Answers: 1

Chemistry, 23.06.2019 03:00
What happens in the particles of a gas when the gas is compressed
Answers: 1
You know the right answer?
The following results were obtained when each of a series of standard silver solutions was analyzed...
Questions

Mathematics, 13.09.2020 16:01

Mathematics, 13.09.2020 16:01

English, 13.09.2020 16:01

Mathematics, 13.09.2020 16:01

Mathematics, 13.09.2020 16:01

English, 13.09.2020 16:01

Mathematics, 13.09.2020 16:01

Mathematics, 13.09.2020 16:01

Mathematics, 13.09.2020 16:01

English, 13.09.2020 16:01

Chemistry, 13.09.2020 16:01

English, 13.09.2020 16:01

Mathematics, 13.09.2020 16:01

Geography, 13.09.2020 16:01

History, 13.09.2020 16:01

Mathematics, 13.09.2020 16:01

Mathematics, 13.09.2020 16:01

Biology, 13.09.2020 16:01

Mathematics, 13.09.2020 16:01

English, 13.09.2020 16:01