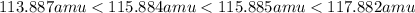The two naturally occurring isotopes of bromine are
81br (80.916 amu, 49.31%) and
79br (78.918 amu, 50.69%).
the two naturally occurring isotopes of chlorine are
37cl (36.966 amu, 24.23%) and
35cl (34.969 amu, 75.77%).
bromine and chlorine combine to form bromine monochloride, brcl.
what are the masses of the four different brcl molecules? express the masses in atomic mass units using six significant figures, in decreasing numeric order (highest to lowest), separated by commas.

Answers: 1
Another question on Chemistry

Chemistry, 21.06.2019 22:30
Which statement best describes the oxidation numbers of the atoms found in magnesium chloride? a. magnesium has a 2- oxidation number and chlorine has a 1+ oxidation number. b. magnesium has a 2- oxidation number and chlorine has a 2+ oxidation number. c. magnesium has a 2+ oxidation number and chlorine has a 1- oxidation number. d. magnesium has a 1+ oxidation number and chlorine has a 1- oxidation number.
Answers: 2

Chemistry, 22.06.2019 05:50
What happens when the temperature of a solution increases?
Answers: 2


Chemistry, 22.06.2019 13:10
Select the correct answer a modure consists of glucose and water. what is the percent composition of glucose in the mixture if it contains 1.3 moles of glucose (cho total mass of the mature is 276 grams? ) and the a 1775
Answers: 1
You know the right answer?
The two naturally occurring isotopes of bromine are
81br (80.916 amu, 49.31%) and
<...
81br (80.916 amu, 49.31%) and
<...
Questions


Computers and Technology, 31.07.2019 00:30



Computers and Technology, 31.07.2019 00:30

Mathematics, 31.07.2019 00:30

Computers and Technology, 31.07.2019 00:30

Mathematics, 31.07.2019 00:30

Computers and Technology, 31.07.2019 00:30


Computers and Technology, 31.07.2019 00:30


Computers and Technology, 31.07.2019 00:30

Biology, 31.07.2019 00:30


Computers and Technology, 31.07.2019 00:30


Computers and Technology, 31.07.2019 00:30


Computers and Technology, 31.07.2019 00:30

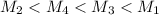
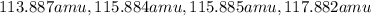
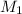 = 80.916 amu + 36.966 amu
= 80.916 amu + 36.966 amu 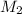 = 78.918 amu + 34.969 amu
= 78.918 amu + 34.969 amu 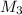 = 80.916 amu + 34.969 amu
= 80.916 amu + 34.969 amu 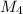 = 78.918 amu + 36.966 amu
= 78.918 amu + 36.966 amu