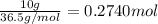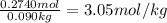Chemistry, 22.01.2020 05:31 jholbrook7643
Calculate the molarity of a 10.0% (by mass) aqueous solution of hydrochloric acid.

Answers: 3
Another question on Chemistry

Chemistry, 22.06.2019 05:30
What royal scientist used the 29th day of frozen vapor to encounter elements for mastering new culinary creations?
Answers: 1

Chemistry, 22.06.2019 11:40
Which of these expressions are correct variations of the combined gas law? p1v1t2 = p2v2t1 both
Answers: 2

Chemistry, 22.06.2019 13:50
Read the chemical equation. 2c2h2 + 5o2 → 4co2 + 2h2o which of the following statements would be correct if one mole of c2h2 was used in this reaction? one mole of oxygen was used in this reaction. five moles of oxygen were used in this reaction. four moles of carbon dioxide were produced from this reaction. two moles of carbon dioxide were produced from this reaction.
Answers: 3

Chemistry, 22.06.2019 14:30
For the reaction shown, find the limiting reactant for each of the following initial amounts of reactants. 4al(s)+3o2(g)→2al2o3(s) a) 1 molal, 1 mol o2 b) 4 molal, 2.6 mol o2 c) 16 molal, 13 mol o2 d) 7.4 molal, 6.5 mol o2
Answers: 3
You know the right answer?
Calculate the molarity of a 10.0% (by mass) aqueous solution of hydrochloric acid....
Questions


Mathematics, 27.05.2021 04:40



Mathematics, 27.05.2021 04:40


Mathematics, 27.05.2021 04:40

Physics, 27.05.2021 04:40


Business, 27.05.2021 04:40

Health, 27.05.2021 04:40


English, 27.05.2021 04:40

Mathematics, 27.05.2021 04:40

Mathematics, 27.05.2021 04:40

Mathematics, 27.05.2021 04:40