Answers: 1
Another question on Chemistry

Chemistry, 22.06.2019 02:40
How many liters of hydrogen gas will be produced at stp from the reaction of 7.179×10^23 atoms of magnesium with 54.219g of phosphoric acid (h3po4) the equation is 3mg + 2h3(> mg(po4)2+3h2
Answers: 1

Chemistry, 22.06.2019 15:30
Using the first volume and temperature reading on the table as v1 and t1, solve for the unknown values in the table below. remember to use the rules of significant figures when entering your numeric response.
Answers: 1

Chemistry, 23.06.2019 02:20
In a chemical reaction, the final amount of the products is determined by the a. universal gas law b. law of definite proportions c. air pressure d. temperature e. none of the above me
Answers: 2

Chemistry, 23.06.2019 05:30
For the reaction i2(g)+br2(g)←−→2ibr(g), kc=280 at 150 ∘c. suppose that 0.450 mol ibr in a 2.00-l flask is allowed to reach equilibrium at 150 ∘c. what is the equilibrium concentration of 2ibr, i2, br2
Answers: 1
You know the right answer?
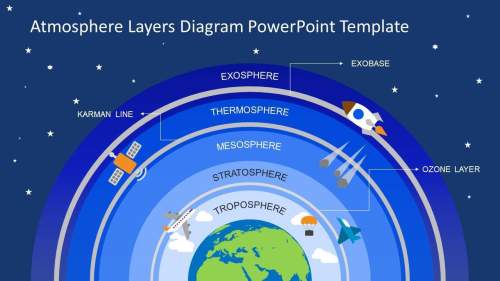
What is the atmospheres in order from earth...
Questions

Mathematics, 26.08.2019 05:50

Mathematics, 26.08.2019 05:50

Mathematics, 26.08.2019 05:50

Mathematics, 26.08.2019 05:50

History, 26.08.2019 05:50



Mathematics, 26.08.2019 05:50


Chemistry, 26.08.2019 05:50

Social Studies, 26.08.2019 05:50

Health, 26.08.2019 05:50

Spanish, 26.08.2019 05:50

Geography, 26.08.2019 05:50





Social Studies, 26.08.2019 05:50