
Reaction of 0.028 g of magnesium with excess hydrochloric acid generated 31.0 ml of hydrogen gas. the gas was collected by water displacement at 22 degree c. the barometric pressure in the lab was 746 mm hg. the vapor pressure of water at 22 degree c is 19.8 mm hg. use dalton's law to calculate the partial pressure of hydrogen gas in the gas-collecting tube. use the combined gas law to calculate the volume of hydrogen at stp. what is the theoretical number of moles of hydrogen that can be produced from 0.028g of mg?

Answers: 1
Another question on Chemistry

Chemistry, 21.06.2019 20:30
Which sentence best describes the formation of igneous rock? a- lava on the surface dries up and makes arock b_melted rocks cools and forms crystals c_rocks under tremendous heat and pressure d_magma is melted rock underground
Answers: 1

Chemistry, 22.06.2019 16:30
How many moles of sulfuric acid (h2so4) are needed to react completely with 6.8 moles of lithium hydroxide (lioh)? 2lioh + h2so4 → li2so4 + 2h2o a. 3.4 mol h2so4b. 6.8 mol h2so4 c. 10.2 mol h2so4 d. 13.6 mol h2so4
Answers: 3

Chemistry, 22.06.2019 23:30
The ammonia molecule in the diagram has the observed bond orientation because
Answers: 1

Chemistry, 23.06.2019 03:30
Mr. rose asked his student to draw a quadrilateral with four unequal sides. an example of this kind of quadrilateral
Answers: 1
You know the right answer?
Reaction of 0.028 g of magnesium with excess hydrochloric acid generated 31.0 ml of hydrogen gas. th...
Questions


History, 04.11.2019 22:31


English, 04.11.2019 22:31



English, 04.11.2019 22:31


Computers and Technology, 04.11.2019 22:31

Mathematics, 04.11.2019 22:31



Chemistry, 04.11.2019 22:31

Health, 04.11.2019 22:31

English, 04.11.2019 22:31

Mathematics, 04.11.2019 22:31

Mathematics, 04.11.2019 22:31



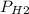 "
"
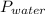
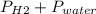
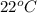 and 726.2 mm Hg and than convert it to STP conditions.
and 726.2 mm Hg and than convert it to STP conditions.
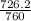
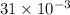 Litre
Litre
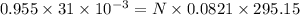
 moles
moles
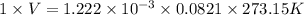
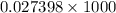 (as 1 L = 1000 ml)
(as 1 L = 1000 ml)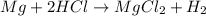
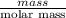
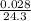
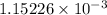 moles
moles



