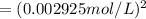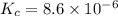Chemistry, 20.01.2020 21:31 amandanunnery33
Titanium(iv) chloride decomposes to form titanium and chlorine, like this: (l)(s)(g) at a certain temperature, a chemist finds that a reaction vessel containing a mixture of titanium(iv) chloride, titanium, and chlorine at equilibrium has the following composition: compound amount calculate the value of the equilibrium constant for this reaction. round your answer to significant digits.

Answers: 1
Another question on Chemistry

Chemistry, 22.06.2019 02:30
Which element forms an ionic bond with flourine? 1) fluorine 2) carbon 3) potassium 4) oxygen
Answers: 1


Chemistry, 22.06.2019 06:00
One does not belong why? ice, gold ,wood ,diamond and table salt
Answers: 1

Chemistry, 22.06.2019 08:40
For each of the following compounds, write the formula then predict whether it would be a strong, weak, or non-electrolyte when placed in di water. for the ionic compounds only, put (s) or (aq) after the forrmula formula strong, weak or non electrolyte? a calcium hydroxide b. silver carbonate c. lead(ii) sulfate d. phosphorus trifluoride e. sodium phosphide f barium sulfate g. strontium acetate h. zinc nitrate
Answers: 3
You know the right answer?
Titanium(iv) chloride decomposes to form titanium and chlorine, like this: (l)(s)(g) at a certain t...
Questions


Mathematics, 28.05.2021 01:30

Mathematics, 28.05.2021 01:30




Mathematics, 28.05.2021 01:30

Mathematics, 28.05.2021 01:30






Arts, 28.05.2021 01:30

Mathematics, 28.05.2021 01:30


English, 28.05.2021 01:30

History, 28.05.2021 01:30

Mathematics, 28.05.2021 01:30

Mathematics, 28.05.2021 01:30

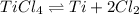
 4.18 g
4.18 g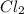 1.08g
1.08g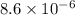 the value of the equilibrium constant for this reaction.
the value of the equilibrium constant for this reaction.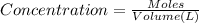
![[TiCl_4]=\frac{4.18 g}{190 g/mol\times 5.2 L}=0.004231 mol/L](/tpl/images/0463/1030/e0fe7.png)
![[Ti]=\frac{1.32 g}{48 g/mol\times 5.2 L}=0.005288 mol/L](/tpl/images/0463/1030/9e0bf.png)
![[Cl_2]=\frac{1.08 g}{71 g/mol\times 5.2 L}=0.002925 mol/L](/tpl/images/0463/1030/2b02a.png)
![K_c=[Cl_2]^2](/tpl/images/0463/1030/ae7ed.png)