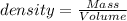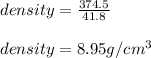Chemistry, 20.01.2020 20:31 mel2001love
Determining density and using density to determine volume or mass
(a) calculate the density of mercury if 1.00 × 10 g occupies a volume of 7.36 cm
(b) calculate the volume of 65.0 g of liquid methanol (wood alcohol) if its density is 0.791 g/ml.
(c) what is the mass in grams of a cube of gold (density = 19.32 g/cm) if the length of the cube is 2.00 cm?
(d) calculate the density of a 374.5-g sample of copper if it has a volume of 41.8 cm a student needs 15.0 g of ethanol for an experiment. if the density of ethanol is 0.789 g/ml, how many milliliters of ethanol are needed? what is the mass, in grams, of 25.0 ml of mercury (density = 13.6 g/ml)?

Answers: 3
Another question on Chemistry

Chemistry, 22.06.2019 00:00
1) these are barrel shaped microtubules in most animal cells, that organize the spindles during cell division
Answers: 1

Chemistry, 22.06.2019 18:30
You open a can of soda at room temperature and hear a hiss. which of the following factors has changed inside the container? a.) atmospheric pressure b.) temperature of gas c.) type of gas d.) amount of gas
Answers: 1

Chemistry, 22.06.2019 23:30
With the largest atoms and the smallest number of valence electrons and with the smallest atoms and the greatest number of valence electrons are the most reactive. a. nonmetals; metals b. nonmetals; transition elements c. transition elements; metals d. metals; nonmetals
Answers: 3

Chemistry, 23.06.2019 00:30
Which of the following best describes technology a. something created for only scientists to use b.the method of thinking that scientists use. c.the application of engineering to create useful products. c. a scientific idea
Answers: 1
You know the right answer?
Determining density and using density to determine volume or mass
(a) calculate the dens...
(a) calculate the dens...
Questions

Chemistry, 16.10.2019 21:30




Health, 16.10.2019 21:30

Mathematics, 16.10.2019 21:30


History, 16.10.2019 21:30





Mathematics, 16.10.2019 21:30

English, 16.10.2019 21:30

Mathematics, 16.10.2019 21:30

Mathematics, 16.10.2019 21:30

Mathematics, 16.10.2019 21:30


English, 16.10.2019 21:30

History, 16.10.2019 21:30

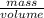
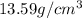 B) volume =
B) volume = 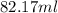 C) mass =
C) mass = 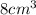 D) density =
D) density = 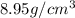
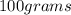
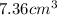
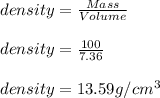
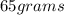
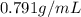
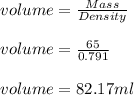
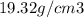
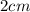
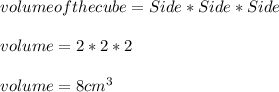
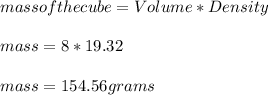 .
.