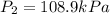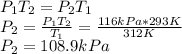The enclosed cabin of a submarine has a volume of 2.4 × 105 liters, a temperature of 312 k, and a pressure of 116 kpa. as people in the cabin breathe, carbon dioxide gas, co2(g), can build up to unsafe levels. air in the cabin becomes unsafe to breathe when the mass of co2(g) in this cabin exceeds 2156 grams. show a numerical setup for calculating the pressure in the submarine cabin if the cabin temperature changes to 293 k

Answers: 3
Another question on Chemistry


Chemistry, 21.06.2019 22:00
Bohr's model could only explain the spectra of which type of atoms? single atoms with one electron single atoms with more than one electron bonded atoms with one electron bonded atoms with more than one electron
Answers: 2


Chemistry, 22.06.2019 21:30
What is another way to determine mass times acceleration?
Answers: 1
You know the right answer?
The enclosed cabin of a submarine has a volume of 2.4 × 105 liters, a temperature of 312 k, and a pr...
Questions



Mathematics, 20.11.2020 22:30



Computers and Technology, 20.11.2020 22:30

Chemistry, 20.11.2020 22:30



Chemistry, 20.11.2020 22:30

Geography, 20.11.2020 22:30

History, 20.11.2020 22:30

Mathematics, 20.11.2020 22:30

Mathematics, 20.11.2020 22:30

Biology, 20.11.2020 22:30



Mathematics, 20.11.2020 22:30

Social Studies, 20.11.2020 22:30