
Consider the following reaction:
a + b + c right arrow d
the rate law for this reaction is as follows:
rate = k time fraction [a][c]^2 over [b]^1/2
suppose the rate of the reaction at certain initial concentrations of a, b, and c is 1.12×10-2 m/s.
what is the rate of the reaction if the concentrations of a and c are doubled and the concentration of b is tripled?
rate 2 = ? m/s

Answers: 3
Another question on Chemistry

Chemistry, 21.06.2019 21:10
How is 0.00235 expressed in proper scientific notation? a. 2.35 × 10-3 b. 0.235 × 10-2 c. 2.35 d. 2.35 × 103
Answers: 1



Chemistry, 23.06.2019 06:00
What does it mean for something to be dissolved in watera- it is submerged in water moleculesb-it is stirred in the water moleculesc- it is surrounded by water molecules d-it has water molecules added to it
Answers: 2
You know the right answer?
Consider the following reaction:
a + b + c right arrow d
the rate law for...
a + b + c right arrow d
the rate law for...
Questions




Mathematics, 14.07.2019 18:40

Chemistry, 14.07.2019 18:40

Mathematics, 14.07.2019 18:40

Health, 14.07.2019 18:40




Spanish, 14.07.2019 18:40

Geography, 14.07.2019 18:40



Mathematics, 14.07.2019 18:40

Mathematics, 14.07.2019 18:40

Mathematics, 14.07.2019 18:40

English, 14.07.2019 18:40

Biology, 14.07.2019 18:40

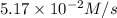
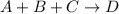
![\text{Rate}_1=k\frac{[A][C]^2}{[B]^{1/2}}](/tpl/images/0459/9288/92a79.png)
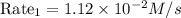
![\text{Rate}_2=k\frac{[2A][2C]^2}{[3B]^{1/2}}\\\\\text{Rate}_2=\frac{2\times 2^2}{3^{1/2}}\times (k\frac{[A][C]^2}{[B]^{1/2}})\\\\\text{Rate}_2=4.62\times (\text{Rate}_1)\\\\\text{Rate}_2=4.62\times 1.12\times 10^{-2}\\\\\text{Rate}_2=5.17\times 10^{-2}M/s](/tpl/images/0459/9288/30eea.png)


