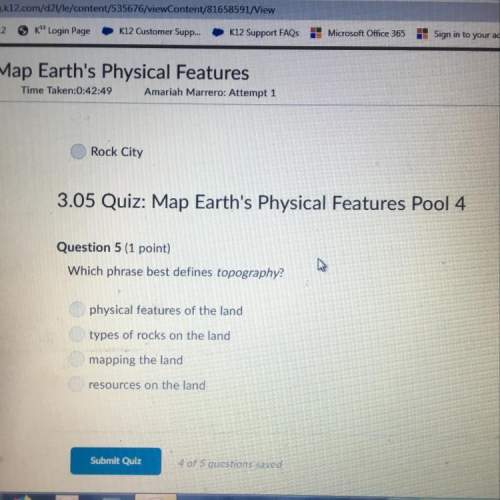
Which of the following is true about the kinetic molecular theory?
(a) the volume of a gas particle is considered to be small – about 0.10 ml.
(b) pressure is due to the collisions of the gas particles with the walls of the container.
(c) gas particles repel each other, but do not attract one another.
(d) adding an ideal gas to a closed container will cause an increase in temperature.
(e) at least two of the above statements are correct.

Answers: 2
Another question on Chemistry


Chemistry, 22.06.2019 06:00
Calculate the mass of silver needed to react with chlorine to produce 126g if silver chloride?
Answers: 3


Chemistry, 22.06.2019 12:20
Consider the reaction of a(g) + b(g) + c(g) => d(g) for which the following data were obtained: experiment initial [a], mol/l initial [b], mol/l initial [c], mol/l initial rate, mol/l.s 1 0.0500 0.0500 0.0100 6.25 x 10^-3 2 0.100 0.0500 0.0100 2.50 x 10^-2 3 0.100 0.100 0.0100 1.00 x 10^-1 4 0.0500 0.0500 0.0200 6.25 x 10^-3 what is the rate law for the reaction?
Answers: 3
You know the right answer?
Which of the following is true about the kinetic molecular theory?
(a) the volume of a...
(a) the volume of a...
Questions



History, 03.06.2020 14:03

Arts, 03.06.2020 14:03


Mathematics, 03.06.2020 14:03

Mathematics, 03.06.2020 14:03



Mathematics, 03.06.2020 14:03

Mathematics, 03.06.2020 14:03

Mathematics, 03.06.2020 14:03


Mathematics, 03.06.2020 14:03

History, 03.06.2020 14:03

Biology, 03.06.2020 14:03

Mathematics, 03.06.2020 14:03


Mathematics, 03.06.2020 14:03

Social Studies, 03.06.2020 14:03