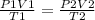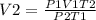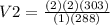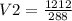Ascuba diver 200 m underwater experiences a temperature of 15 oc and a pressure of
2 atm if sh...

Ascuba diver 200 m underwater experiences a temperature of 15 oc and a pressure of
2 atm if she takes a large breath of oxygen (inhaling 2.0 l) and then swims to the
surface without exhaling, what will be the volume of oxygen in her lungs? the
temperature at the surface is 30 oc and the pressure is 1 atm

Answers: 1
Another question on Chemistry

Chemistry, 22.06.2019 04:40
*will mark you brainliest + 15 points ** why does the equilibrium of a system shift when the pressure is increased? a. to maximize the stress on the system b. to stop restoring equilibrium to the system c. to increase the total moles of gas in the system d. to decrease the total moles of gas in the system
Answers: 3

Chemistry, 22.06.2019 06:00
Calculate - analysis of compound composed of iron and oxygen yields 174.86 of fe and 75.14g of o. what is the empirical formula for this compound?
Answers: 3

Chemistry, 22.06.2019 07:40
21. consider the following chemical reaction: n2+ o2 2 no if 10.0 g of n2 reacts with excess oxygen then how many grams of no can be formed? a) 10.7 g b) 21.4 g c) 32.9 g d) 42.8 g page 4 of 8
Answers: 2

Chemistry, 22.06.2019 17:10
Increasing the substrate concentration in an enzymatic reaction could overcome which of the following? a) the need for a coenzymeb) allosteric inhibitionc) competitive inhibitiond) insufficient cofactors
Answers: 1
You know the right answer?
Questions

Mathematics, 24.09.2019 16:10




Social Studies, 24.09.2019 16:10

Mathematics, 24.09.2019 16:10



English, 24.09.2019 16:10

Chemistry, 24.09.2019 16:10

History, 24.09.2019 16:10

English, 24.09.2019 16:10



World Languages, 24.09.2019 16:10

Biology, 24.09.2019 16:10




Social Studies, 24.09.2019 16:10