
Chemistry, 13.01.2020 20:31 juansoto227711
Which of the following represents the ground state electron configuration for the mn3 ion?

Answers: 1
Another question on Chemistry

Chemistry, 22.06.2019 16:50
Answer asap need by wednesday morning calculate the ph of 0.16m ch3cooh which has ka = 1.74 x 10-5 mol dm-3 best answer will be brainliest
Answers: 3

Chemistry, 23.06.2019 06:40
The combustion of methane, ch4, releases 890.4kj/mol. that is, when one mole of methane is burned,890.4 kj are given off to the surroundings. this meansthat the products have 890.4 kj less than the reactants.thus, ah for the reaction = - 890.4 kj. a negative symbolforah indicates an exothermic reaction.ch (g) + 20 (g)> co2 (g) + 2 h0 (1); ah = - 890.4 kga) how much energy is given off when 2.00 mol of ch,are burned? b) how much energy is released when 22.4g of ch. areburned?
Answers: 1

Chemistry, 23.06.2019 09:00
20 grams of water. she poured out 15 grams. which of the following physical properties of the water changes? a .boiling point b. density c .electrical conductivity d .volume
Answers: 2

You know the right answer?
Which of the following represents the ground state electron configuration for the mn3 ion?...
Questions


Physics, 05.11.2020 23:00



English, 05.11.2020 23:00

Mathematics, 05.11.2020 23:00


Engineering, 05.11.2020 23:00

History, 05.11.2020 23:00



Mathematics, 05.11.2020 23:00


Mathematics, 05.11.2020 23:00

Mathematics, 05.11.2020 23:00

Mathematics, 05.11.2020 23:00


Engineering, 05.11.2020 23:00

Mathematics, 05.11.2020 23:00

History, 05.11.2020 23:00

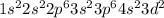
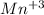 has 22 electrons arround the nucleous. Its ground state configuration following the diagonal rule is:
has 22 electrons arround the nucleous. Its ground state configuration following the diagonal rule is: ![[Ar] 4s^2 3d^2](/tpl/images/0453/2485/866a9.png)


