
Calculate the voltage of the cell.
ag(s)|agbr(s), nabr(aq, 1.0 m)||cdcl2(aq, 0.050 m)|cd(s)
agbr(s) + e- ⇌ ag(s) + br-
eo = 0.071 v
ag+ + e- ⇌ ag(s)
eo = 0.799 v
cd2+ + 2e- ⇌ cd(s)
eo = -0.402 v
ksp (agbr(s)) = 5.0 × 10-13
a. -0.678 v
b. -0.511 v
c. -0.423 v
d. 0.511 v

Answers: 1
Another question on Chemistry


Chemistry, 22.06.2019 12:00
Which of the following units is not an official si unit? mole liter kilogram ampere
Answers: 1

Chemistry, 22.06.2019 16:10
Given the following equation: 2a1 + 3mgcl2 --> 2alcl3 + 3mg how many moles of aluminum chloride are produced from 2.5 moles of magnesium chloride?
Answers: 1

You know the right answer?
Calculate the voltage of the cell.
ag(s)|agbr(s), nabr(aq, 1.0 m)||cdcl2(aq, 0.050 m)|cd...
ag(s)|agbr(s), nabr(aq, 1.0 m)||cdcl2(aq, 0.050 m)|cd...
Questions

Mathematics, 19.11.2020 21:20

Mathematics, 19.11.2020 21:20

Mathematics, 19.11.2020 21:20



Mathematics, 19.11.2020 21:20

Biology, 19.11.2020 21:20

Chemistry, 19.11.2020 21:20


Spanish, 19.11.2020 21:20

Mathematics, 19.11.2020 21:20

Mathematics, 19.11.2020 21:20


Mathematics, 19.11.2020 21:20

Mathematics, 19.11.2020 21:20

Mathematics, 19.11.2020 21:20

Mathematics, 19.11.2020 21:20


Mathematics, 19.11.2020 21:20

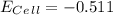
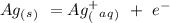 Eo = -0.799 V
Eo = -0.799 V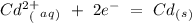 Eo = -0.402 V
Eo = -0.402 V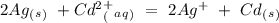 Eo = -1.201
Eo = -1.201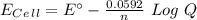
![E_C_e_l_l=E^{\circ}-\frac{0.0592}{n}Log\frac{[Ag^+]^2}{[Cd^2^+]}](/tpl/images/0450/4403/f186a.png)
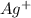 concentration, so:
concentration, so: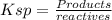
![5x10^-^1^3=[x][1]](/tpl/images/0450/4403/89725.png)
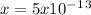
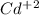 given by the problem we can calculate Q:
given by the problem we can calculate Q:![Q=\frac{[5x10^-^1^3]^2}{[0.05]}=5x10^-^2^4](/tpl/images/0450/4403/d0af4.png)
![E_C_e_l_l=-1.201}-\frac{0.0592}{2}Log[5x10^-^2^4]](/tpl/images/0450/4403/4601d.png)


