
Chemistry, 10.01.2020 06:31 aidanwindsor1738
Asource of zinc metal can be zinc ore containing zinc(ii) sulfide. the ore is roasted in pure oxygen to produce the oxide and then reduced with carbon to form elemental zinc and carbon monoxide. 2 zns + o2 2 zno + 2 so2 zno + c zn + co a crucible containing a sample of 0.50 mol zns was roasted in pure oxygen, then reduced with 1.00 mol carbon. what mass remained in the crucible after cooling?

Answers: 2
Another question on Chemistry


Chemistry, 21.06.2019 17:30
This is a mixture that has the same composition throughout.
Answers: 1

Chemistry, 21.06.2019 22:30
Ionic compounds are made of ions, and yet the overall charge of an ionic compound is neutral. why?
Answers: 1

Chemistry, 22.06.2019 08:30
Which common material is an example of a polymer? (25 pts) a. steel b. plastic c. petroleum d. rubbing alcohol
Answers: 2
You know the right answer?
Asource of zinc metal can be zinc ore containing zinc(ii) sulfide. the ore is roasted in pure oxygen...
Questions




Mathematics, 25.05.2021 21:40

English, 25.05.2021 21:40

Mathematics, 25.05.2021 21:40

Biology, 25.05.2021 21:40

Mathematics, 25.05.2021 21:40


Mathematics, 25.05.2021 21:40

Mathematics, 25.05.2021 21:40

Chemistry, 25.05.2021 21:40

Mathematics, 25.05.2021 21:40

Business, 25.05.2021 21:40


Mathematics, 25.05.2021 21:40

Mathematics, 25.05.2021 21:40

Mathematics, 25.05.2021 21:40

Mathematics, 25.05.2021 21:40

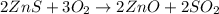 ..[1]
..[1]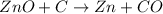 ..[2]
..[2]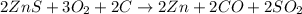 ..[3]
..[3]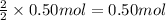 of Zn.
of Zn.

