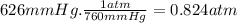What is the volume (l) occupied by a mole of an ideal gas,
ifthe pressure is 626mmhg and the t...


Answers: 1
Another question on Chemistry


Chemistry, 22.06.2019 06:00
Ethanol (c2h5oh) is produced from the fermentation of sucrose in the presence of enzymes. c12h22o11(aq) + h2o(g) 4 c2h5oh(l) + 4 co2(g) determine the theoretical yield and the percent yields of ethanol if 680. g sucrose undergoes fermentation and 326.5 g ethanol is obtained. theoretical _ g _ percent %
Answers: 1

Chemistry, 22.06.2019 12:30
If 22.5 liters of oxygen reacted with excess of hydrogen, how many liters of water vapor could be produced?
Answers: 3

Chemistry, 22.06.2019 15:30
Each of the following reactions is allowed to come to equilibrium and then the volume is changed as indicated. predict the effect (shift right, shift left, or no effect) of the indicated volume change. drag the appropriate items to their respective bins.co(g) + h2o(g) < => co2(g) + h2(g) (volume is decreased) pcl3(g) + cl2(g) < => pcl5(g) (volume is increased) caco3(s)< => cao(s) + co2(g) (volume is increased)
Answers: 1
You know the right answer?
Questions


Social Studies, 30.03.2021 17:30

Mathematics, 30.03.2021 17:30

Mathematics, 30.03.2021 17:30

Mathematics, 30.03.2021 17:30

Mathematics, 30.03.2021 17:30


Mathematics, 30.03.2021 17:30

Health, 30.03.2021 17:30


Mathematics, 30.03.2021 17:30

Mathematics, 30.03.2021 17:30

Mathematics, 30.03.2021 17:30

Mathematics, 30.03.2021 17:30