
Chemistry, 09.01.2020 01:31 zachspencer6444
Given the following information, what is the concentration of h2o(g) at equilibrium? [h2s](eq) = 0.671 m [o2](eq) = 0.587 m kc = 1.35 2h2s(g) + o2(g) ⇌ 2s(s) + 2h2o(g)

Answers: 1
Another question on Chemistry

Chemistry, 21.06.2019 22:30
In order to calculate the amount of heat transferred you must know the __ and specific heat of the material, as well as the change in temperature. a. volume b. density c. mass d. enthalpy
Answers: 1

Chemistry, 22.06.2019 03:40
Kc = 0.040 for the system below at 450oc. if a reaction is initiated with 0.40 mole of cl2 and 0.40 mole of pcl3 in a 2.0 liter container, what is the equilibrium concentration of cl2 in the same system? pcl5(g) ⇄ pcl3(g) + cl2(g)
Answers: 3


Chemistry, 22.06.2019 09:00
Which process does not require the presence of a physical substance in order to transfer heat? air in the atmosphere is heated by the ground. this warm air then rises, and cooler air falls. this is an example of what type of process? how is conduction different from radiation?
Answers: 1
You know the right answer?
Given the following information, what is the concentration of h2o(g) at equilibrium? [h2s](eq) = 0....
Questions



Spanish, 19.04.2020 04:48

Mathematics, 19.04.2020 04:48

Social Studies, 19.04.2020 04:48

Biology, 19.04.2020 04:48


Social Studies, 19.04.2020 04:48







Physics, 19.04.2020 04:48

English, 19.04.2020 04:48


Physics, 19.04.2020 04:48

History, 19.04.2020 04:48

Mathematics, 19.04.2020 04:48

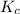
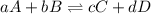
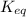 is written as:
is written as:![K_{c}=\frac{[C]^c[D]^d}{[A]^a[B]^b}](/tpl/images/0447/6972/b6f47.png)
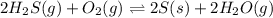
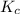 for above equation is:
for above equation is:![K_c=\frac{[H_2O]^2}{[H_2S]^2\times [O_2]}](/tpl/images/0447/6972/73c62.png)
![[H_2S]_{eq}=0.671M](/tpl/images/0447/6972/623cf.png)
![[O_2]_{eq}=0.587M](/tpl/images/0447/6972/4ff7f.png)

![1.35=\frac{[H_2O]^2}{(0.671)^2\times 0.587}](/tpl/images/0447/6972/14791.png)
![[H_2O]=\sqrt{(1.35\times 0.671\times 0.671\times 0.587)}=0.597M](/tpl/images/0447/6972/73db2.png)


